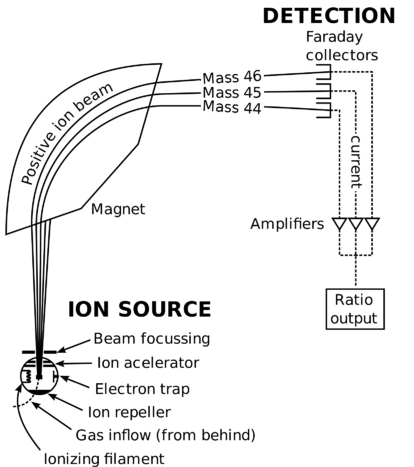
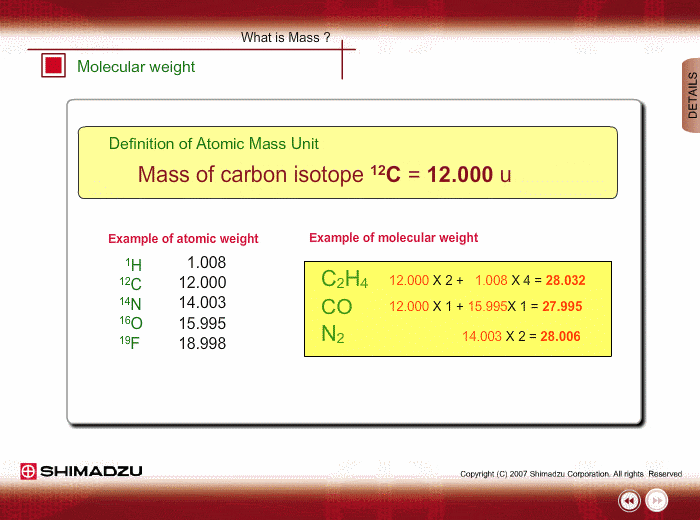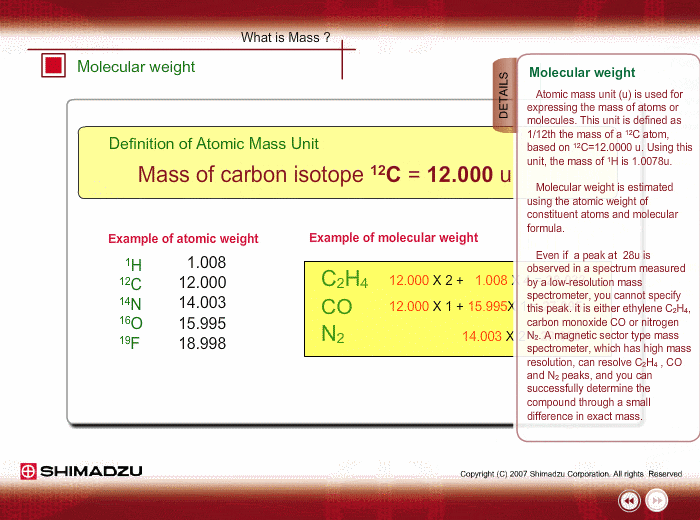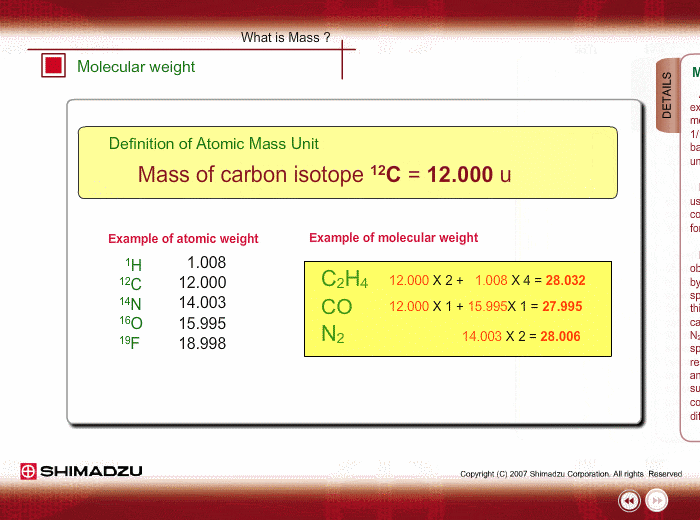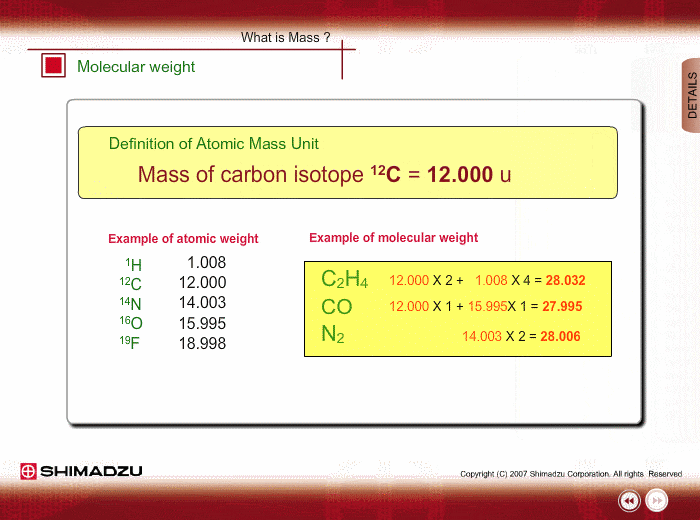
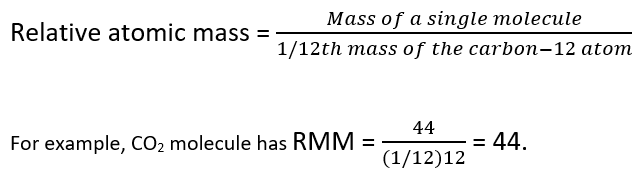What is 1/12 of the mass of a carbon 12 atom? And why do we compare atomic masses of other elements with respect to it? - Quora
What is 1/12 of the mass of a carbon 12 atom? And why do we compare atomic masses of other elements with respect to it? - Quora

if we consider that 1/6, in place of 1/12, mass of carbon atom is taken to be relative atomic mass - Chemistry - Some Basic Concepts of Chemistry - 13772503 | Meritnation.com

Which of the following represents 12 u?1 point(a) Mass of 1 hydrogen atom(b) Mass of C-12 atom km(c) Mass of - Brainly.in

Question Video: Calculating the Atomic Mass of a Fluorine-19 Atom in Unified Atomic Mass Units | Nagwa

SOLVED: if the reference of atomic weight scale is changed from the present one viz, c = 12 unit to c=24 units , will it lead to any change in the atomic
28. If we considered that 1/6 in place of 1/12 mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mole substance will 1) becomes

Assertion A : One atomic mass unit is defined as one twelfth of the mass of one carbon 12 atom.Reason R : Carbon 12 isotope is the most abundant isotope of carbon

If we consider that 1/6 , in place of 1/12 ; mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mole of a substance will