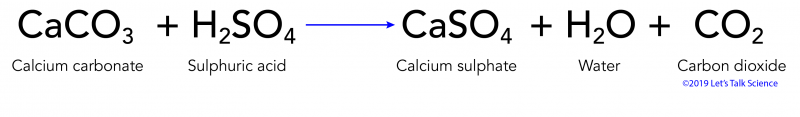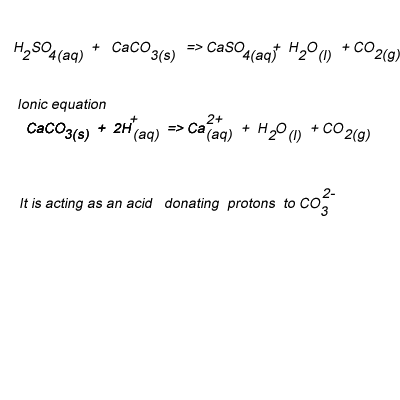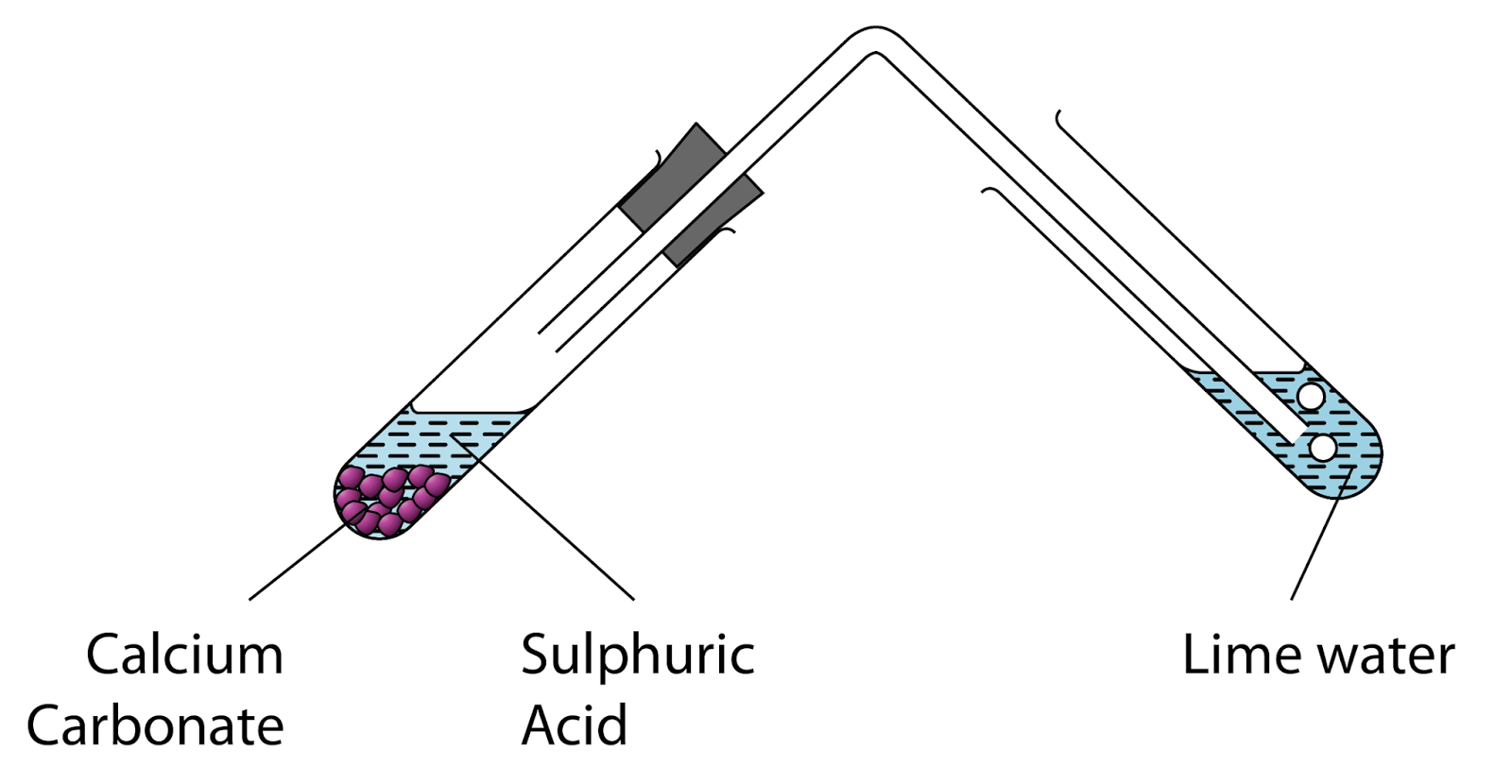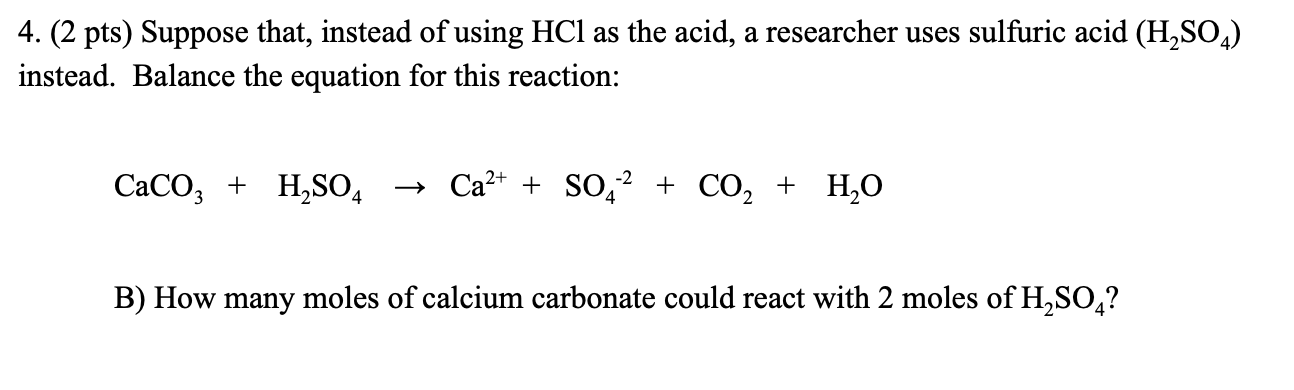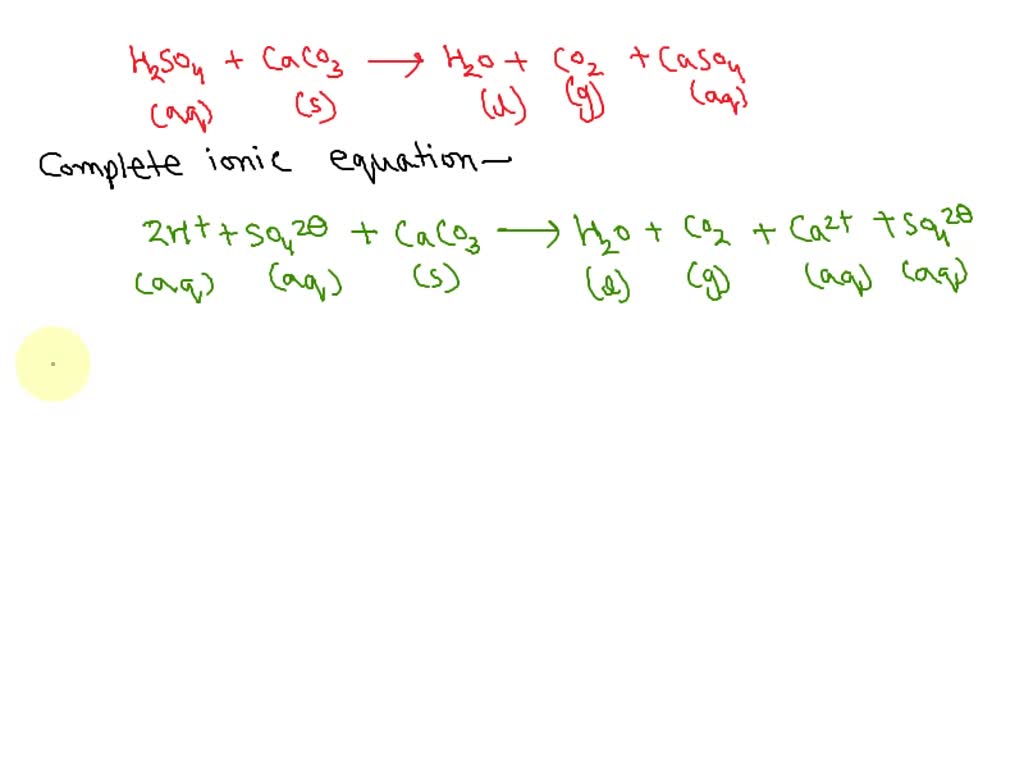
SOLVED: Write complete ionic and net ionic equations for the reaction between sulfuric acid (H2SO4) and calcium carbonate (CaCO3). H2SO4(aq)+CaCO3(s) →H2O(l)+CO2(g)+CaSO4(aq)

Consumption of CO 2 and production of HCO 3 − in silicates and calcium... | Download Scientific Diagram