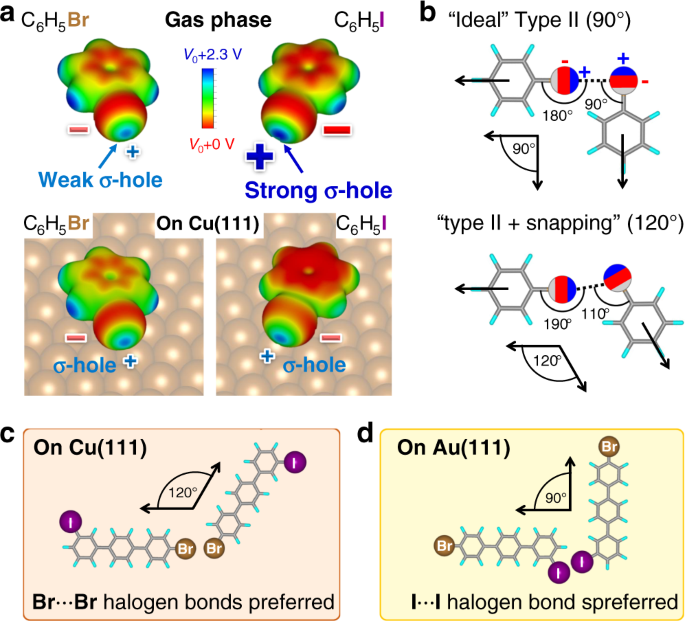
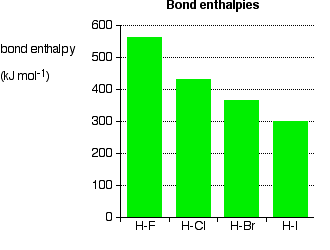
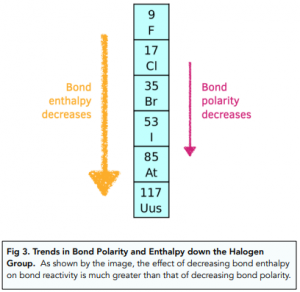
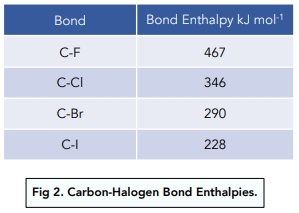
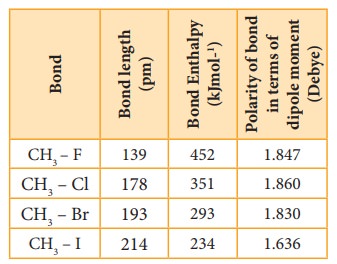
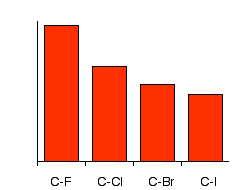
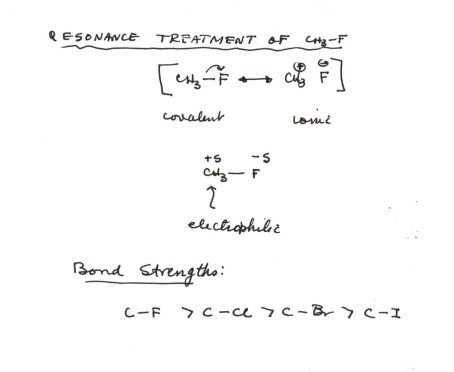
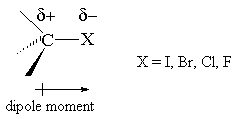244. Dissociation energies of carbon–halogen bonds. The bond strengths allyl–X and benzyl–X - Journal of the Chemical Society (Resumed) (RSC Publishing)
Why doesn't the bond polarity of a carbon halogen bond increase from Chlorine~Bromine~Iodine? - Quora
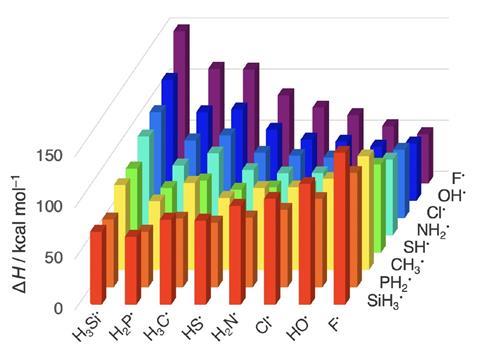
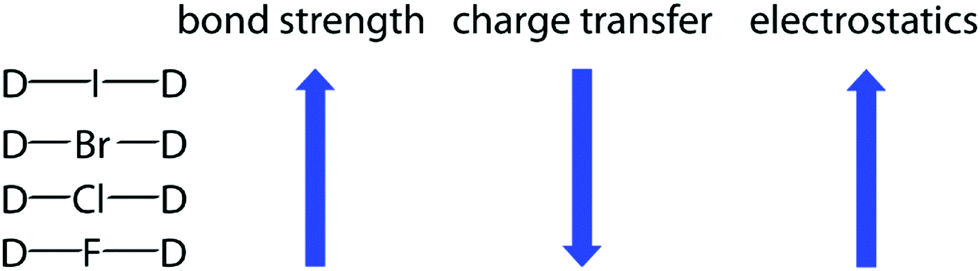
Textbook electronegativity model fails when it comes to carbon–halogen bond strengths | Research | Chemistry World