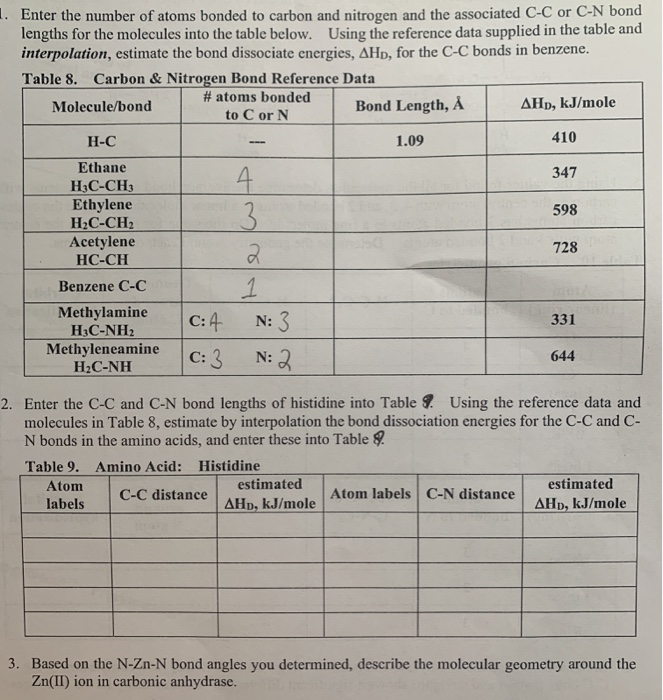
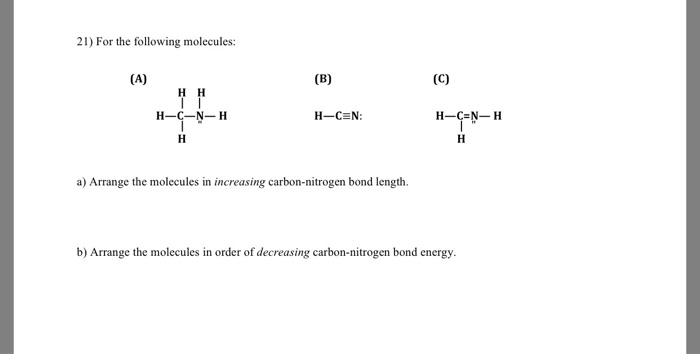
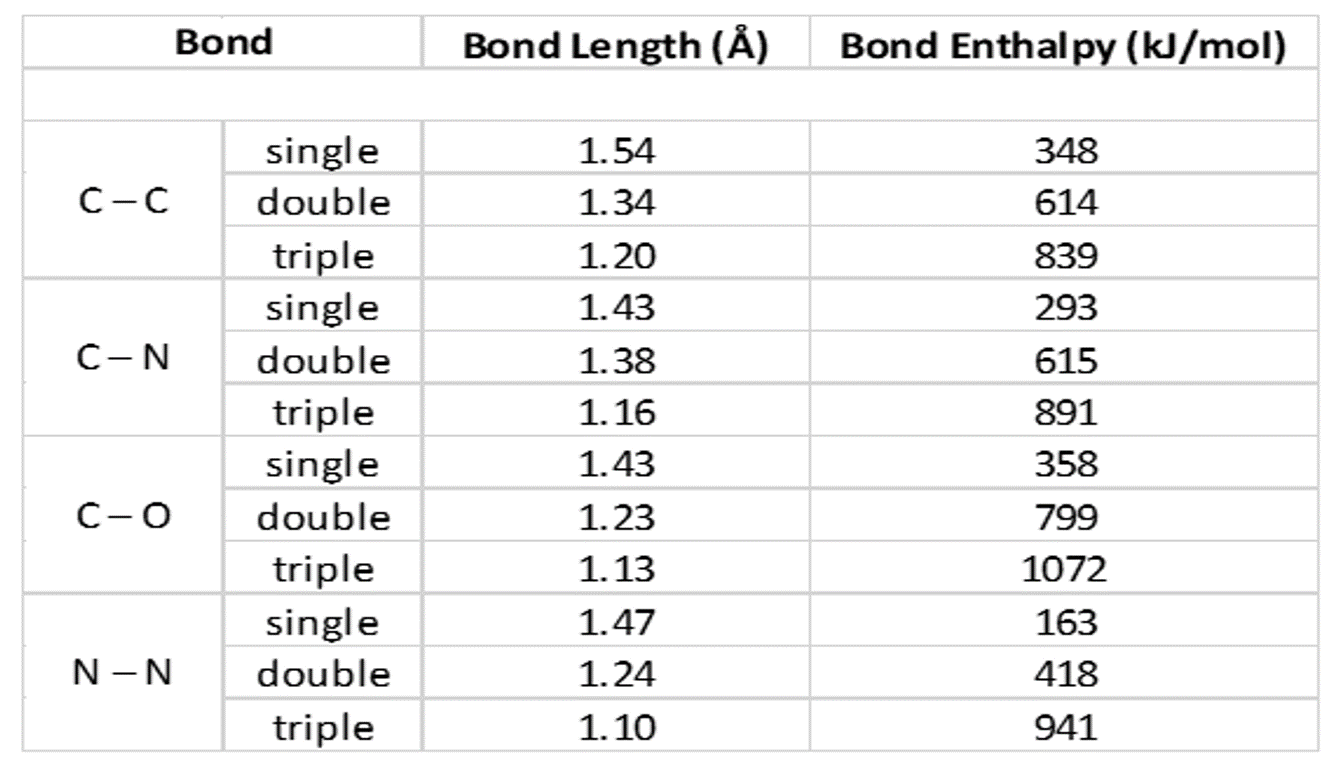
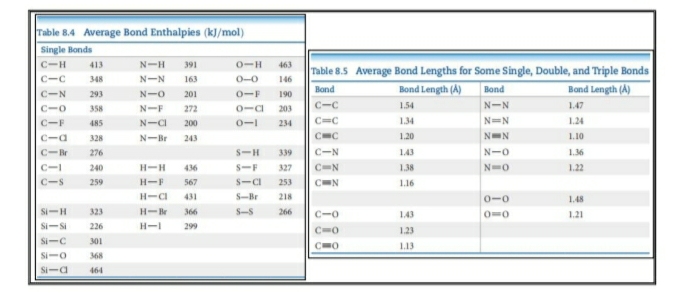
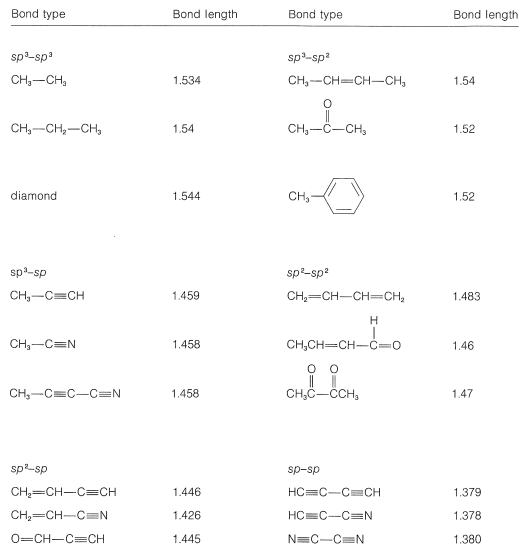
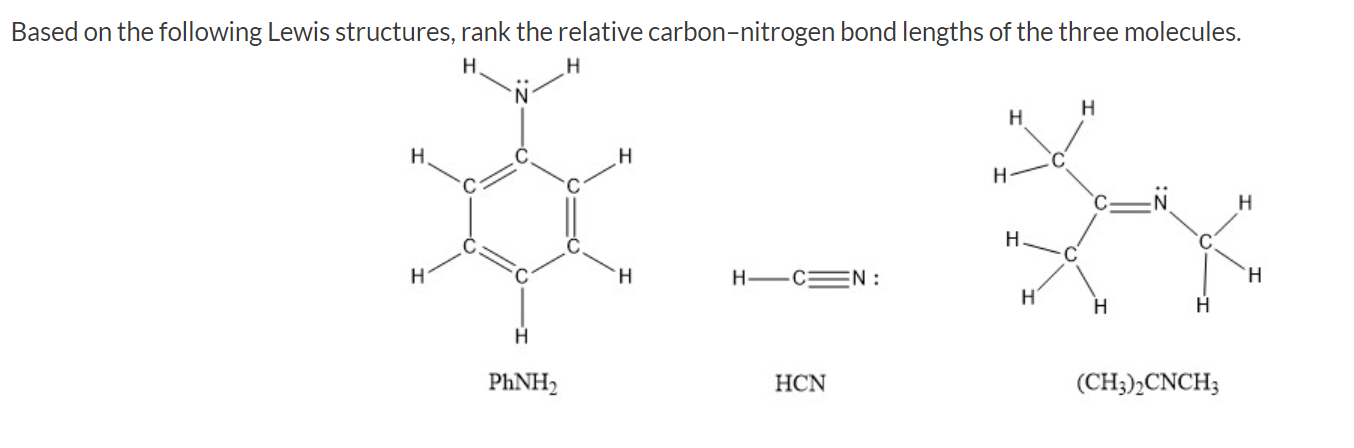
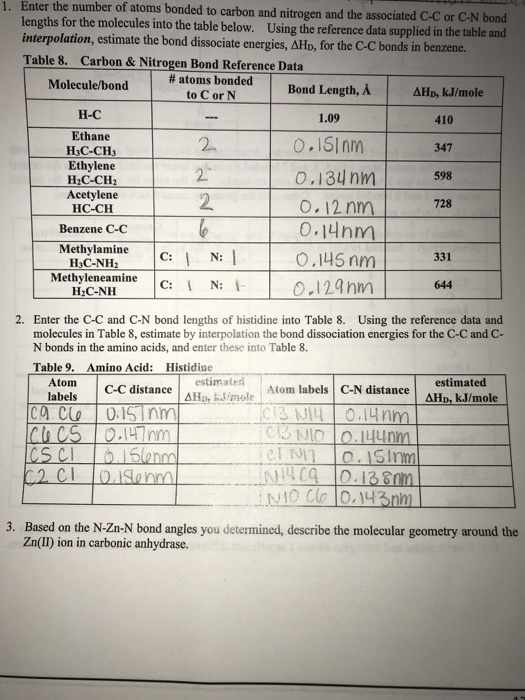
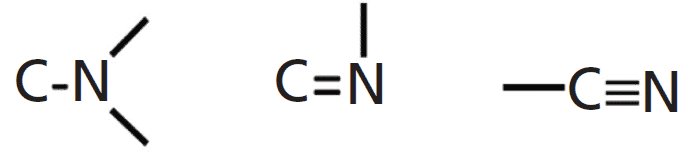SOLVED: Identify the compound with the longest carbon nitrogen bond. CH;CHzCH-NH CH3CHzNHz CH3CHC=N The length of the carbon-nitrogen bonds are the same

Calculated bond lengths of predicted CN structure; a (3 Â 3) supercell... | Download Scientific Diagram
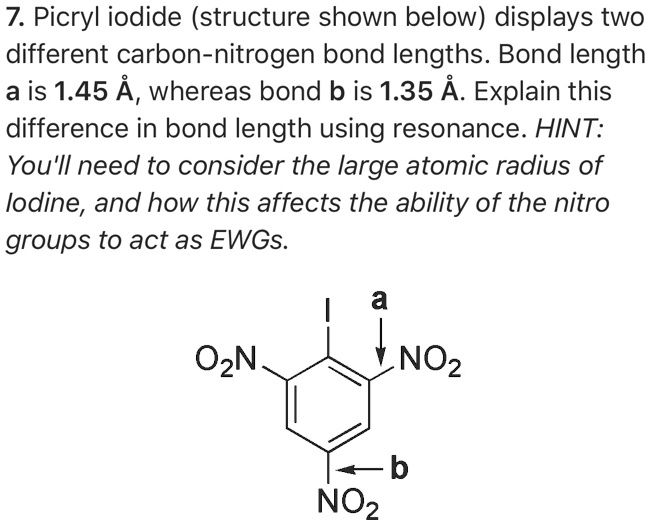
SOLVED: 7. Picryl iodide (structure shown below) displays two different carbon-nitrogen bond lengths. Bond length a is 1.45 A, whereas bond b is 1.35 A. Explain this difference in bond length using