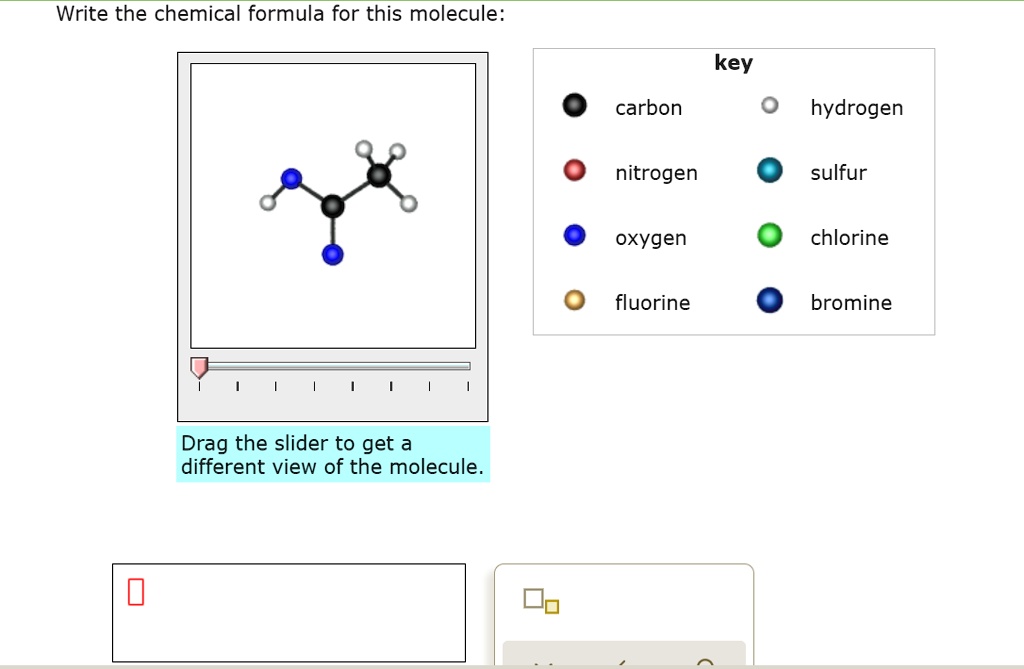
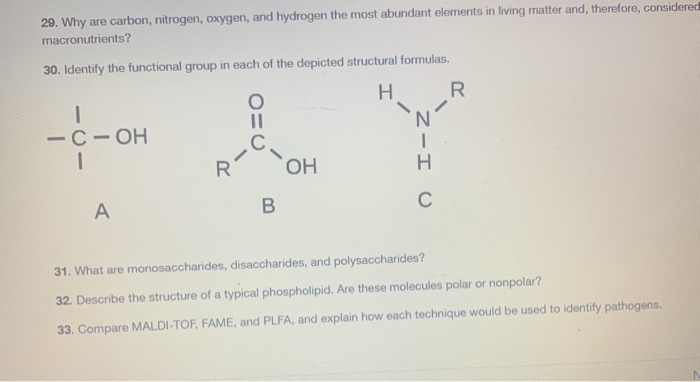
An organic compound contains 20 % carbon, 6.7 % hydrogen, and 46.67 % nitrogen. Its molecular weight was found to be 60 . Find the molecular formula of the compound.

Inorganic Compounds of Carbon, Nitrogen, and Oxygen - Leikauf - Major Reference Works - Wiley Online Library

What is the empirical formula for a compound containing 38.8% carbon, 16.2% hydrogen and 45.1% nitrogen?

Following the rule that each atom of carbon, oxygen, and nitrogen reacts to achieve a complete outer shell of eight valence electrons, add unshared pairs of electrons as necessary to complete the

Calculate the Empirical Formula for a compound with the following composition: 46.16% carbon; 53.84% nitrogen 1)Change % to grams (if needed) 2)Convert. - ppt download

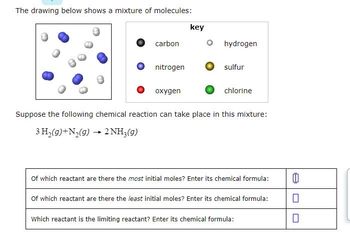
The drawing below shows a mixture of molecules: carbon nitrogen oxygen hydrogen sulfur chlorine Suppose the - Brainly.com