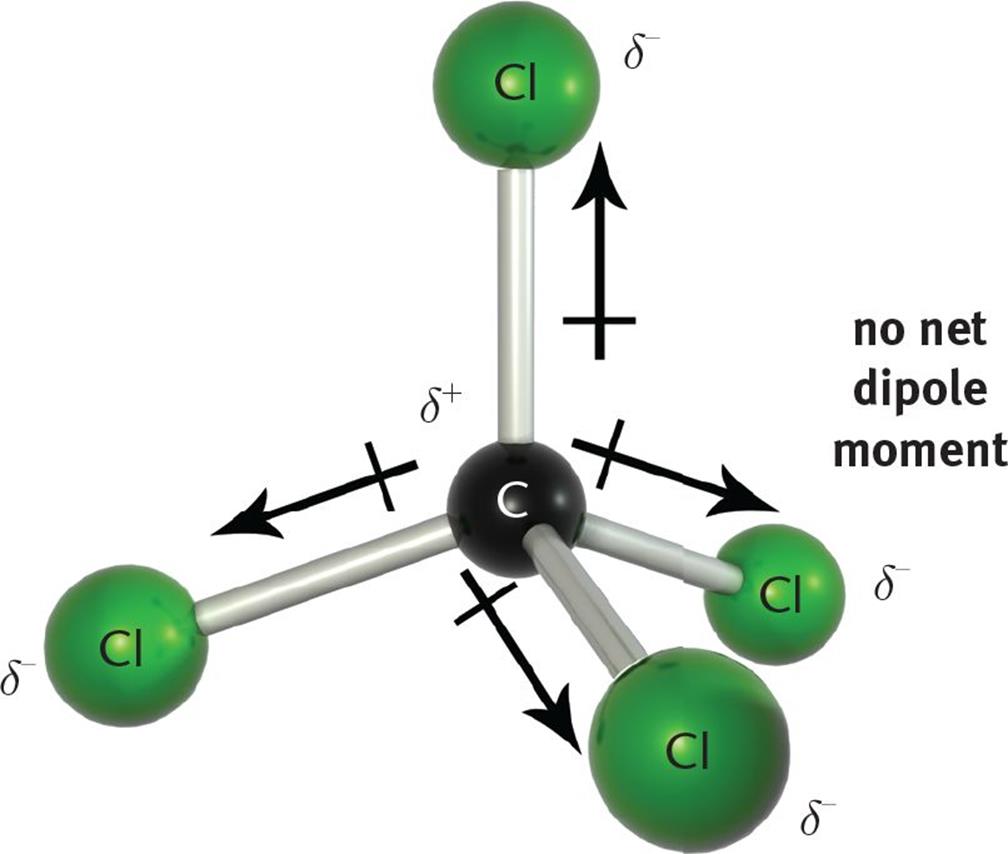
Answer the following:1) Write the formula and draw the electron dot structure of carbon tetrachloride.

Carbon tetrachloride CCl4 lewis dot structure, molecular geometry, polar or nonpolar, Bond angle | Molecular geometry, Molecular shapes, Molecular
Draw the structural formula of carbon tetrachloride and state the type of bond present in it - Sarthaks eConnect | Largest Online Education Community

In the formation of Carbon tetra chloride molecule, state the following.a) Electronic configuration of nearest inert gas attained.b) How many electrons are sahred/ transferred in bond formation?c) Which type of bonds these

can you expain the geometry of carbon tetrachloride,ethane and ethene - Chemistry - Chemical Bonding and Molecular Structure - 3770600 | Meritnation.com

Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar
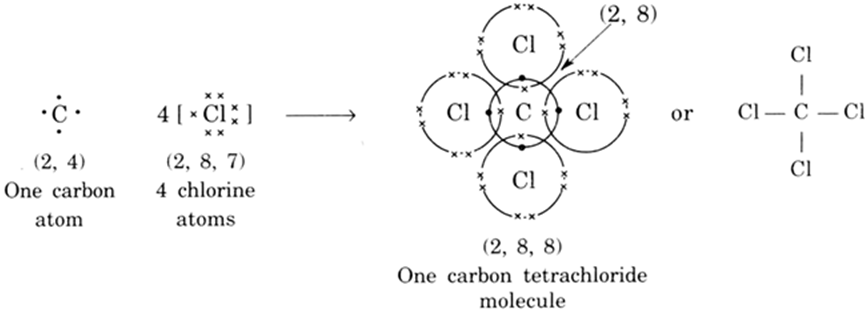
Explain the formation of covalent bonds in (i) chlorine molecule; (ii) carbon tetrachloride and (iii) ammonia. from Science Carbon and its Compounds Class 10 Jammu and Kashmir Board
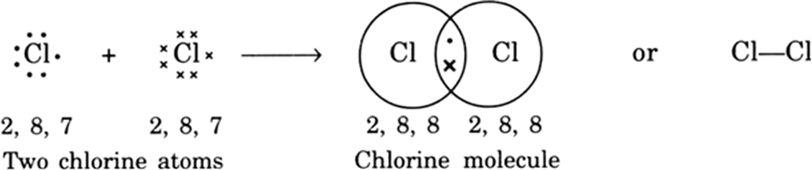
Explain the formation of covalent bonds in (i) chlorine molecule; (ii) carbon tetrachloride and (iii) ammonia. from Science Carbon and its Compounds Class 10 CBSE