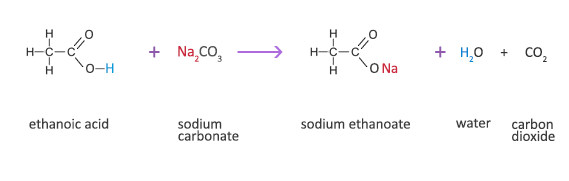
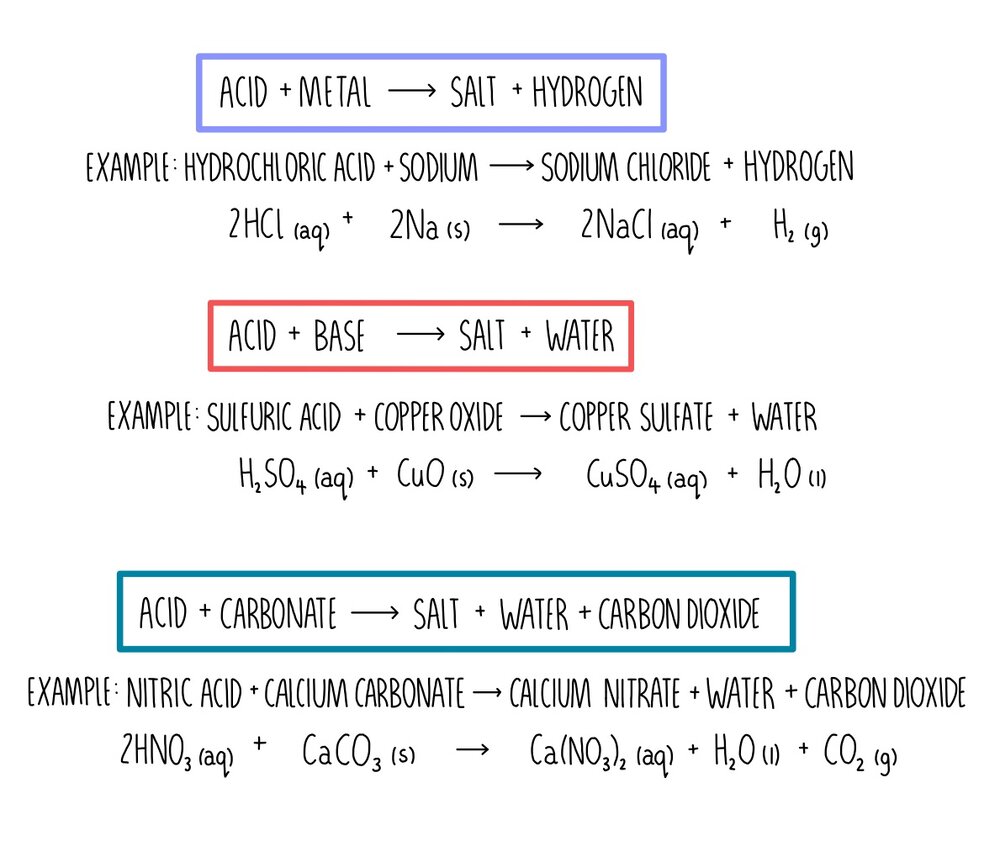
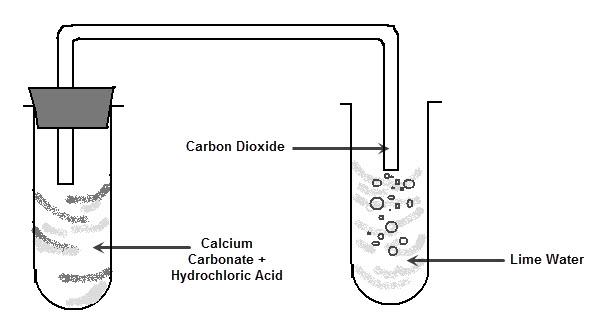
In the reaction between potassium carbonate and hydrochloric acid, 25 grams of salt were produced. If excess acid was available, how many grams of the carbonate were used? - Quora

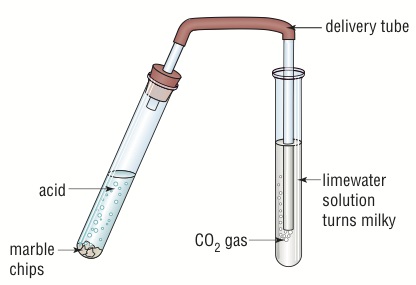
Question Video: Calculating the Average Rate of Reaction of Hydrochloric Acid with Calcium Carbonate | Nagwa

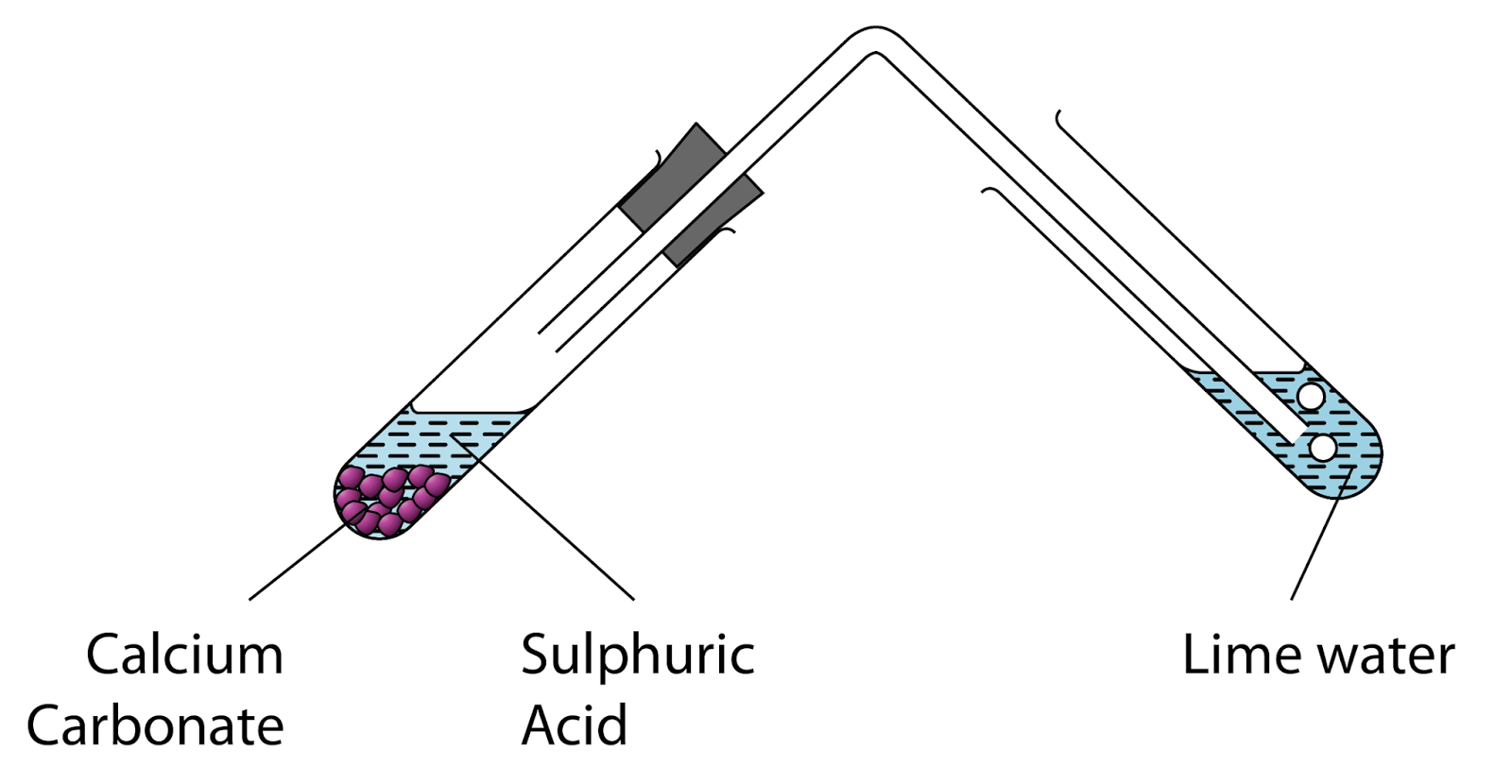
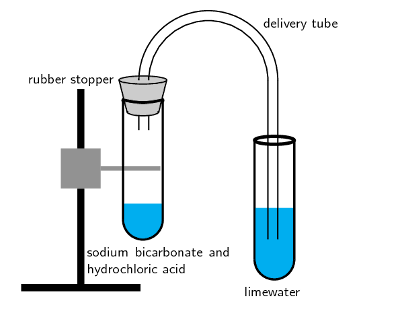
calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide Stock Photo - Alamy

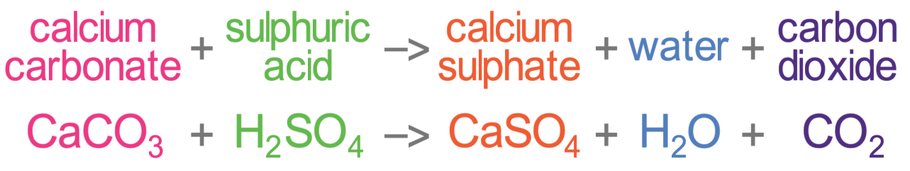
.png)