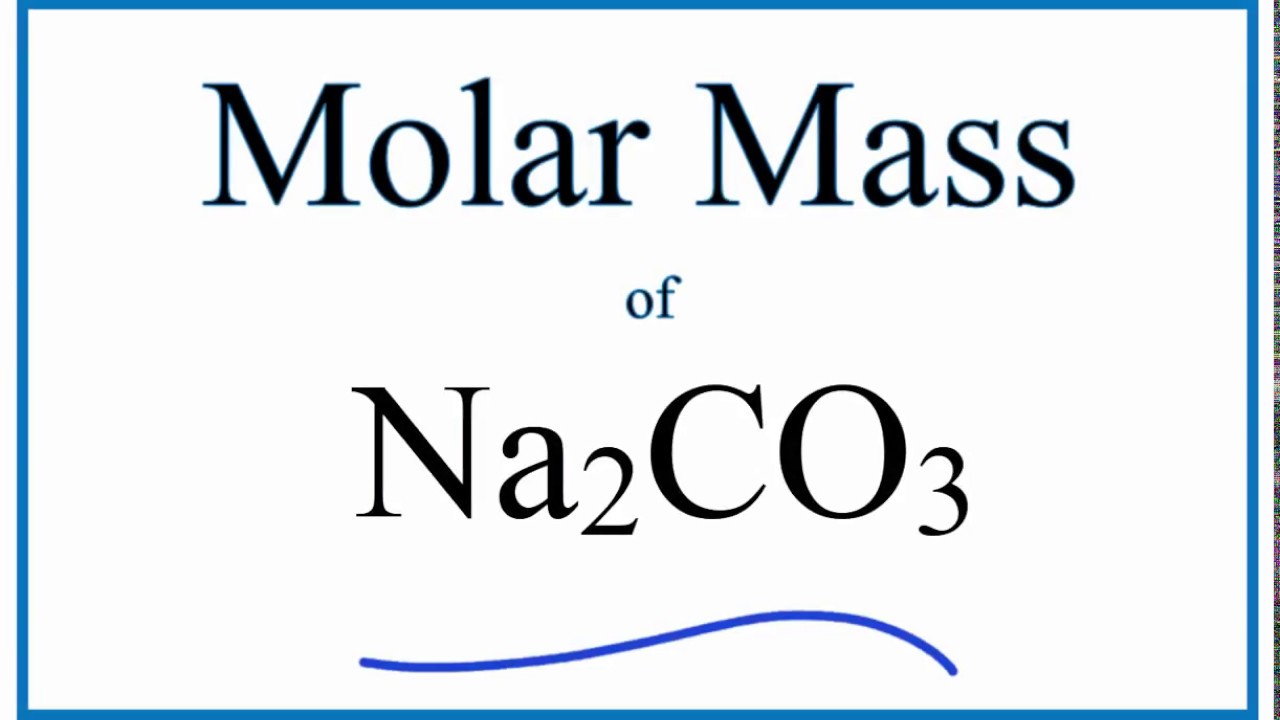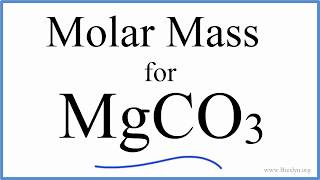Define molecular mass. Calculate molecular mass of Calcium Carbonate, CaCO3. (Given atomic mass of Ca=40 u; - Brainly.in

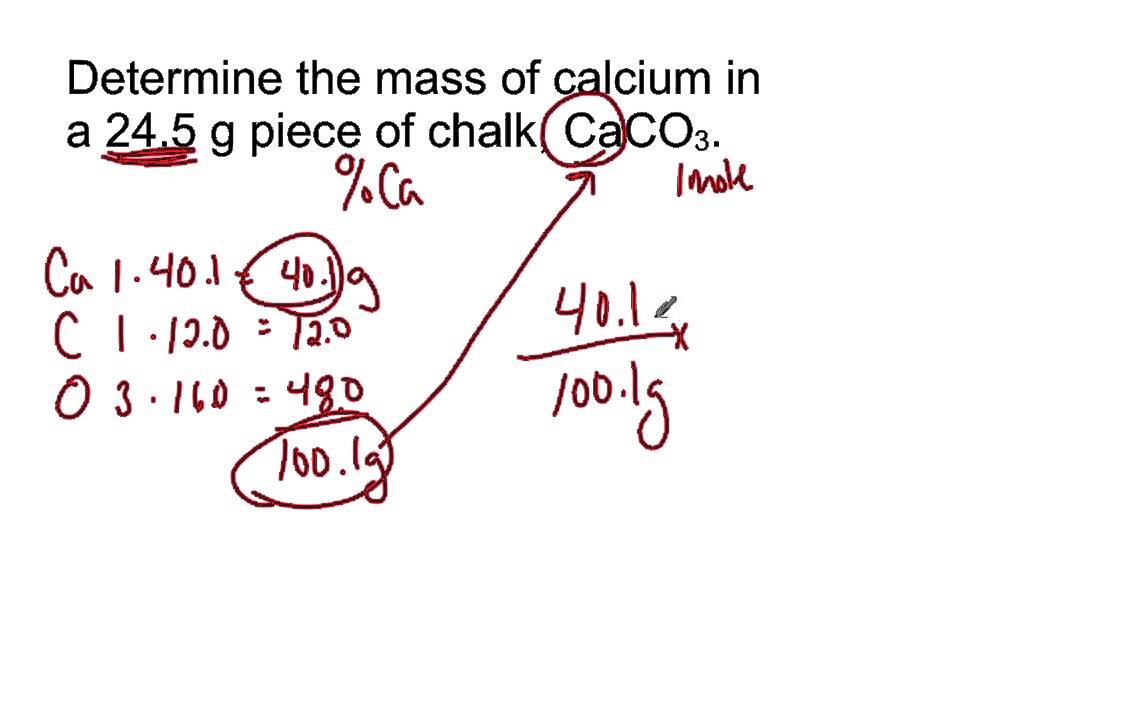
Calculate the mass per cent of each element present in the molecule of calcium carbonate - CBSE Class 9 Science - Learn CBSE Forum

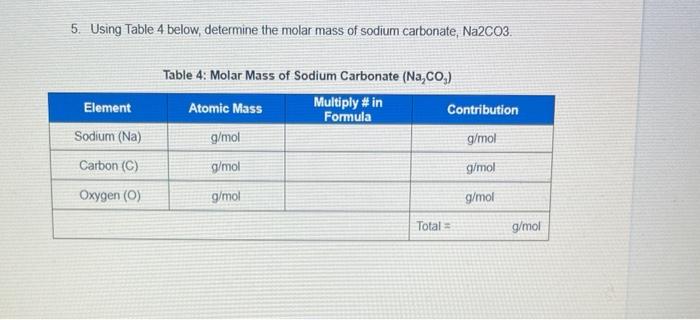
SOLVED: Find the normality of sodium carbonate (molar mass = 106) in a solution containing 0.212 g carbonate in 100 ml solution if the carbonate is used as a dibasic base means it has 2 OH-.
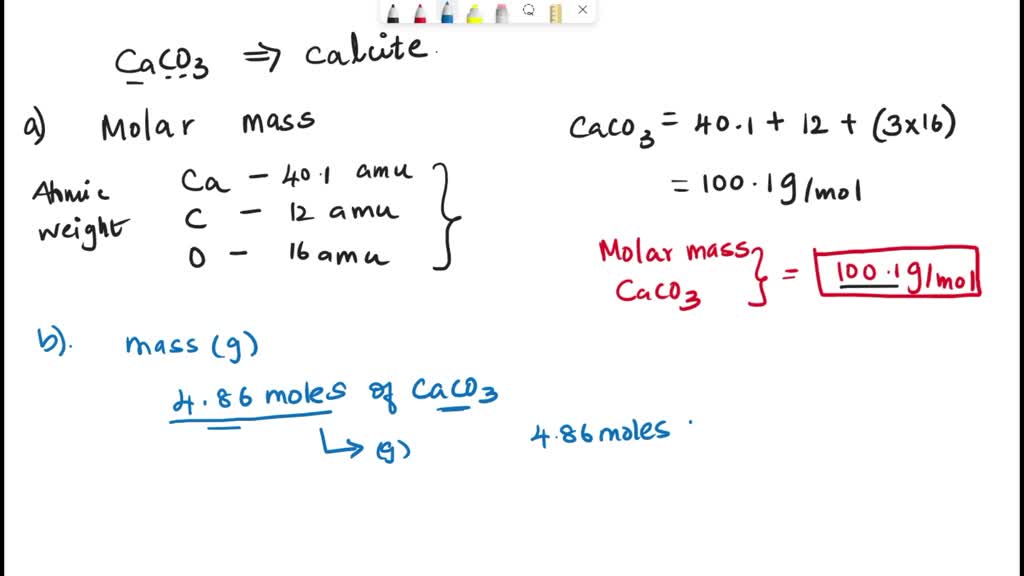
SOLVED: Calcium carbonate (CaCO;), also called calcite: Calculate the molar mass of caleium carbonate. A certain sample of calcium carbonate contains 4.86 moles What is the mass in this sample? grams What
![If the relative molecular mass of ammonium nitrate is 80, the percentage of nitrogen and oxygen in ammonium nitrate is: [N = 14, H = 1, O = 16] If the relative molecular mass of ammonium nitrate is 80, the percentage of nitrogen and oxygen in ammonium nitrate is: [N = 14, H = 1, O = 16]](https://dwes9vv9u0550.cloudfront.net/images/3796715/307077b2-1bb0-4bde-a9d0-fe5f6659fec9.jpg)
If the relative molecular mass of ammonium nitrate is 80, the percentage of nitrogen and oxygen in ammonium nitrate is: [N = 14, H = 1, O = 16]
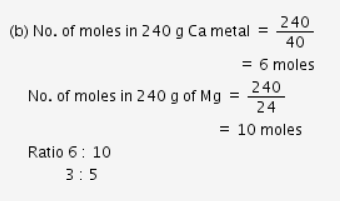
Calculate the molecular mass of CaCO3 (At mass Ca = 40 u, C = 12 u, O = 16 u) - CBSE Class 9 - Learn CBSE Forum


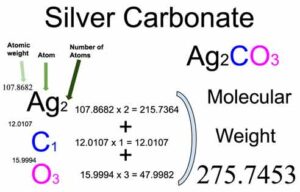
![Barium Carbonate [BaCO3] Molecular Weight Calculation - Laboratory Notes Barium Carbonate [BaCO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/barium-carbonate-molecular-weight-calculation-300x205.jpg)