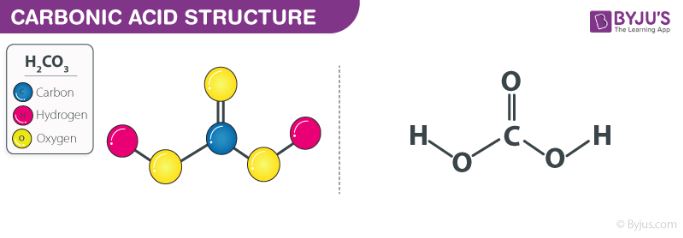
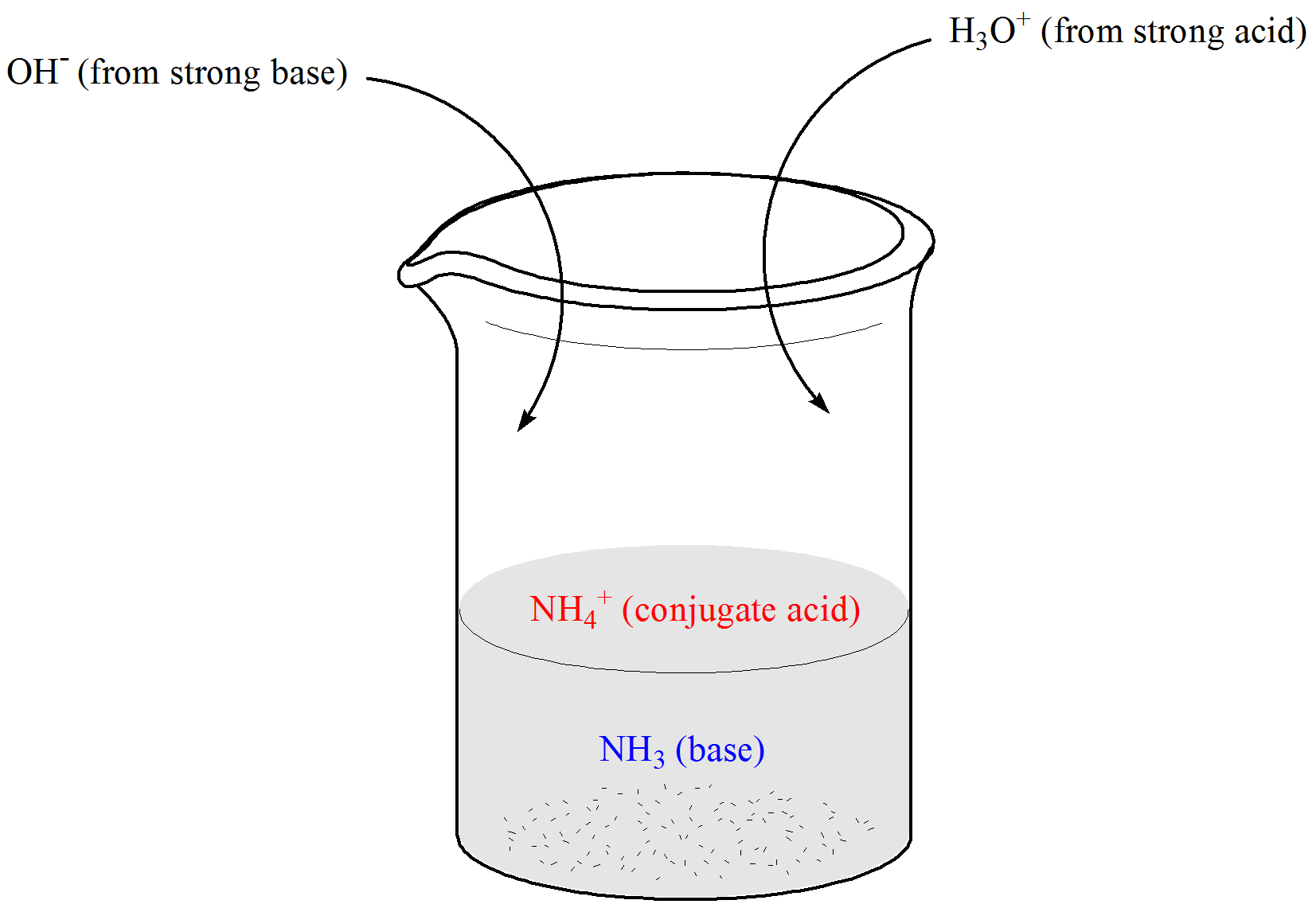
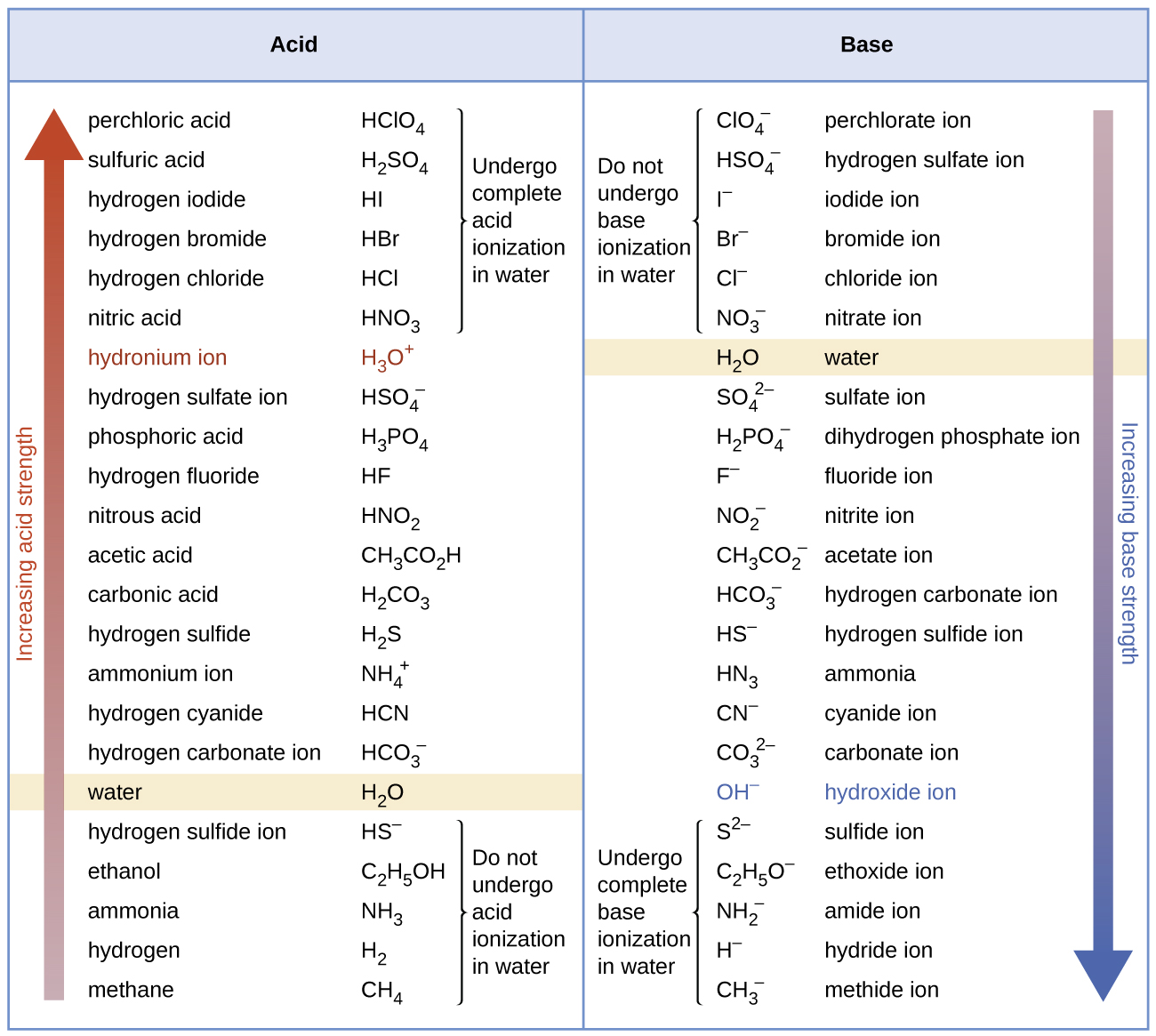
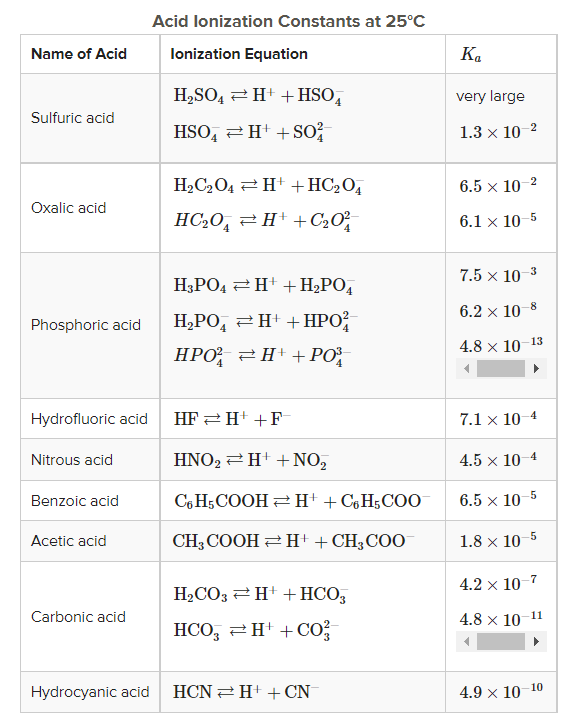
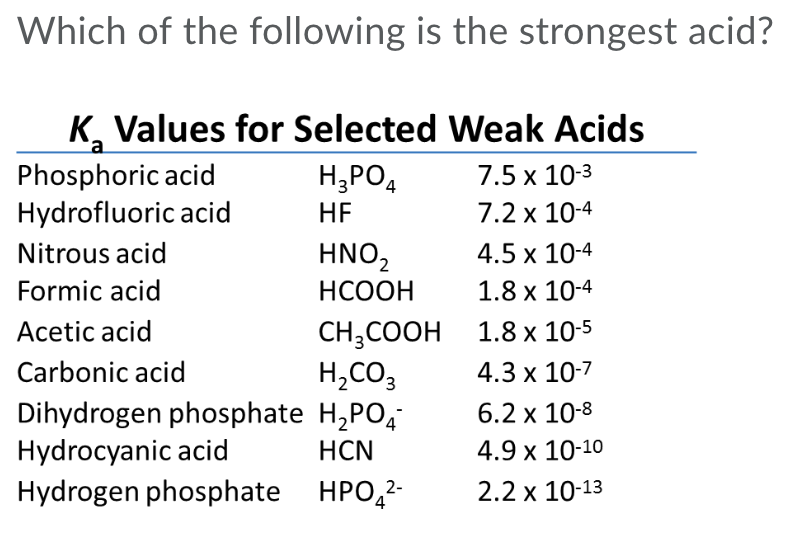
THE CO 2 -H 2 O SYSTEM - I Carbonic acid is a weak acid of great importance in natural waters. The first step in its formation is the dissolution of CO. - ppt download
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora

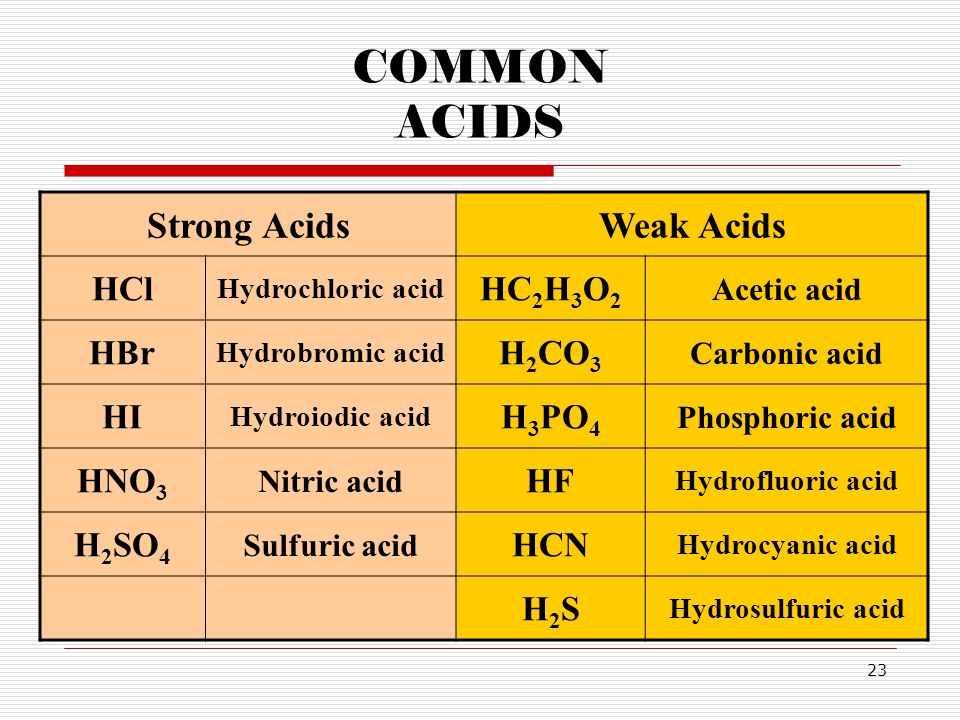
SOLVED: Classify each of the following aS a Strong acid (sa) O a Weak acid (wa) and indicate how each should be written in aqueous solution: Classify In solution this acid should

Acetaminophen is a weaker acid than carbonic acid (H2CO3), but aspirin is a stronger acid than carbonic acid. prepare a flow diagram like the one in this experiment, showing a procedure that