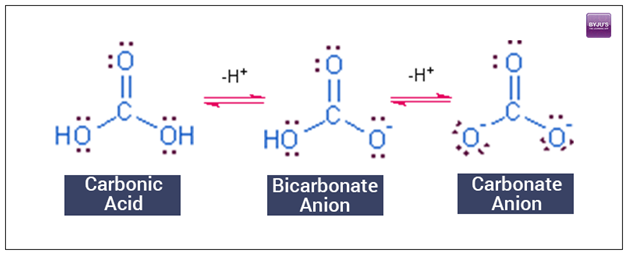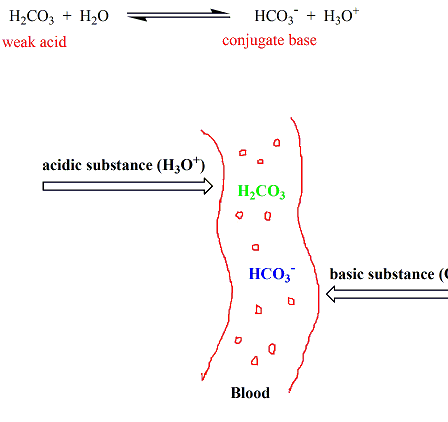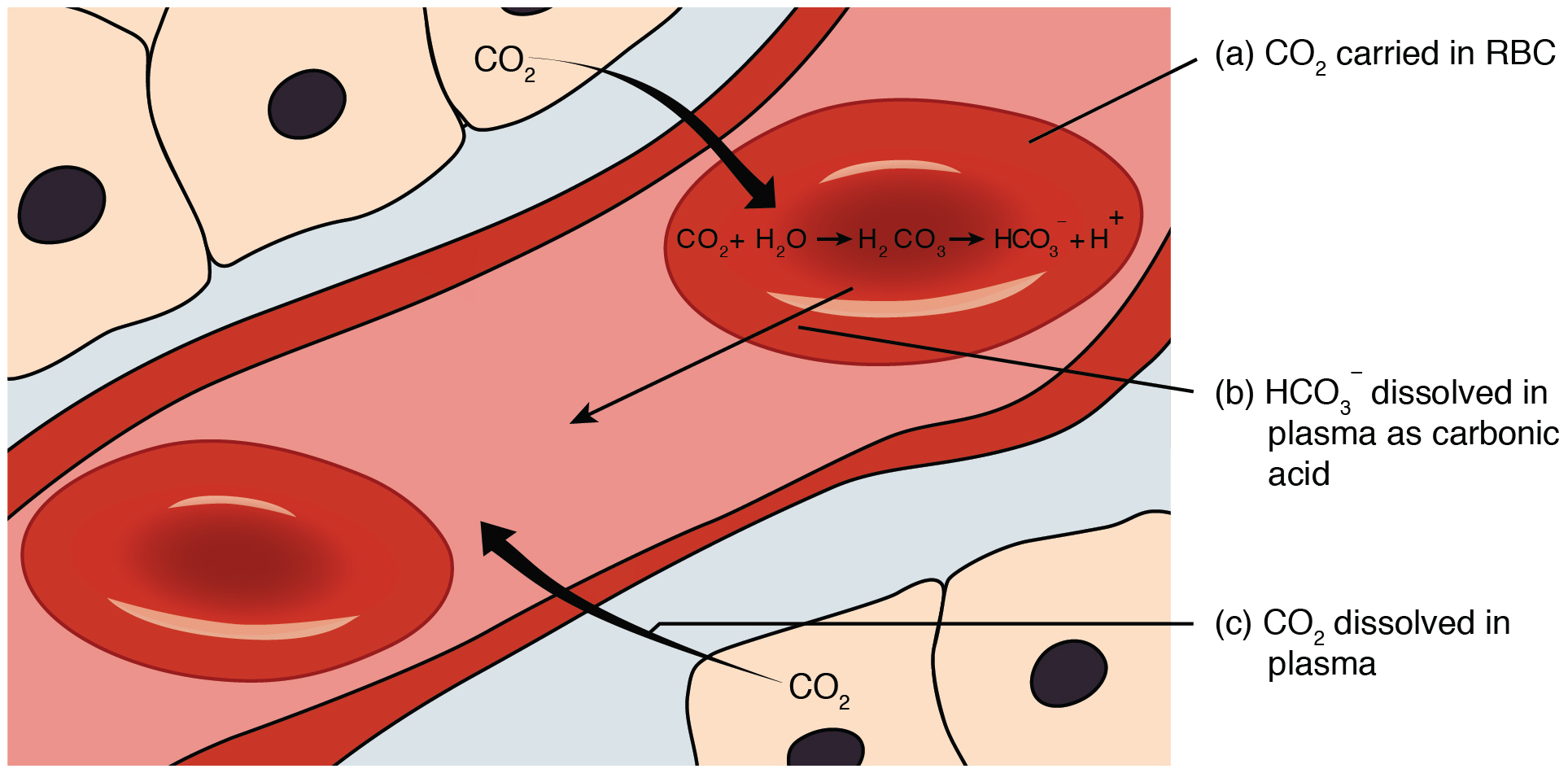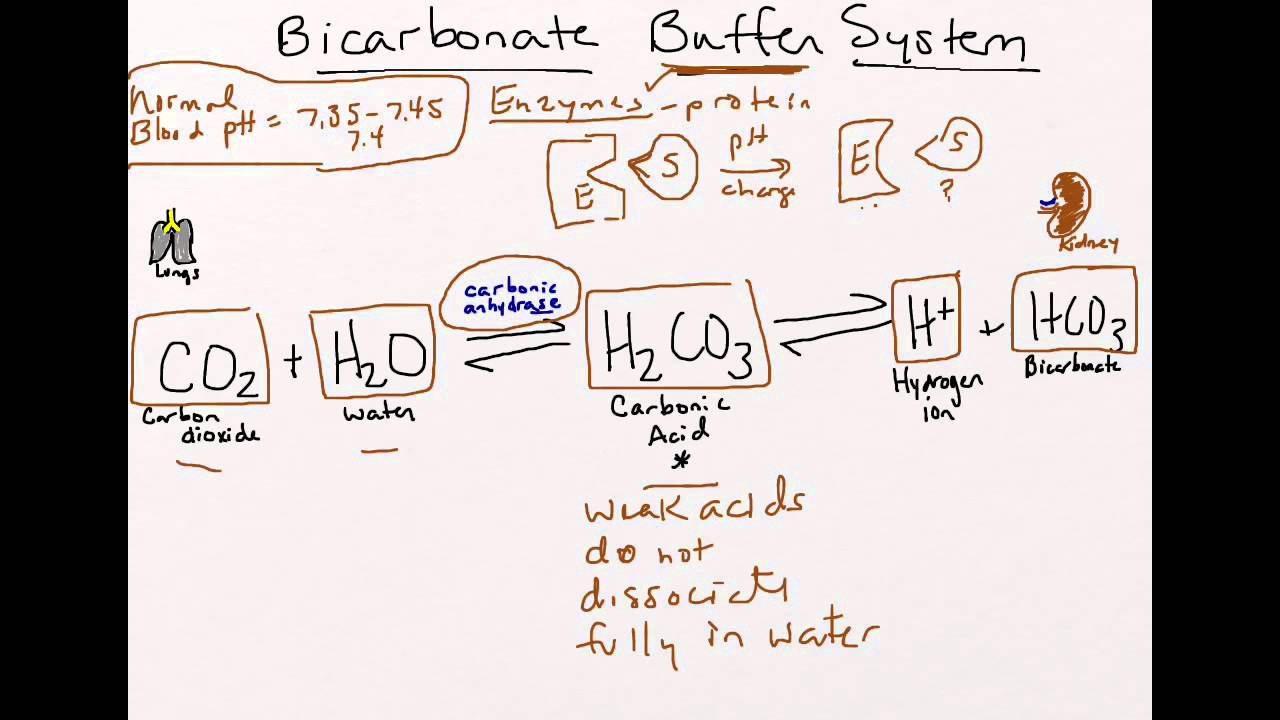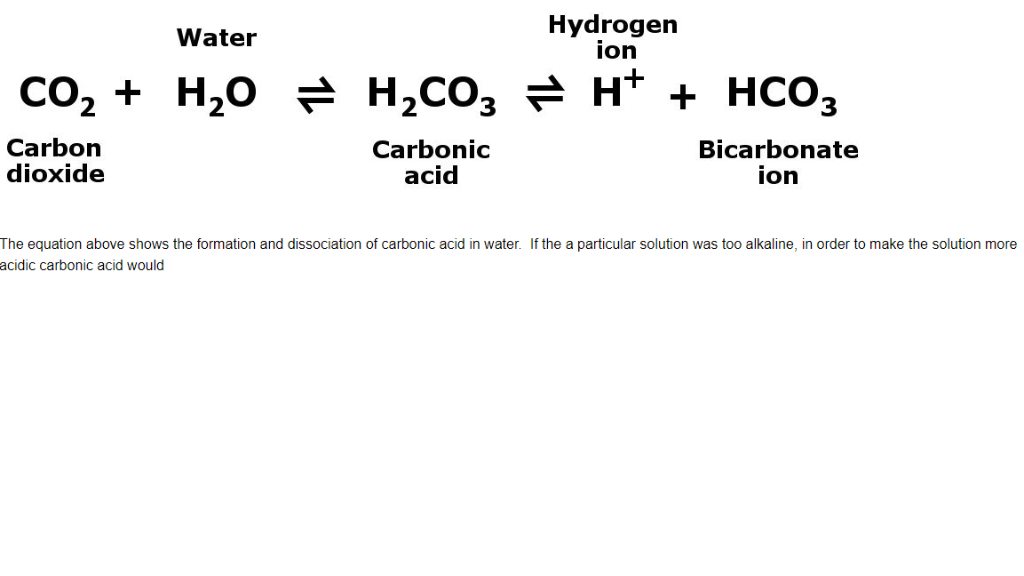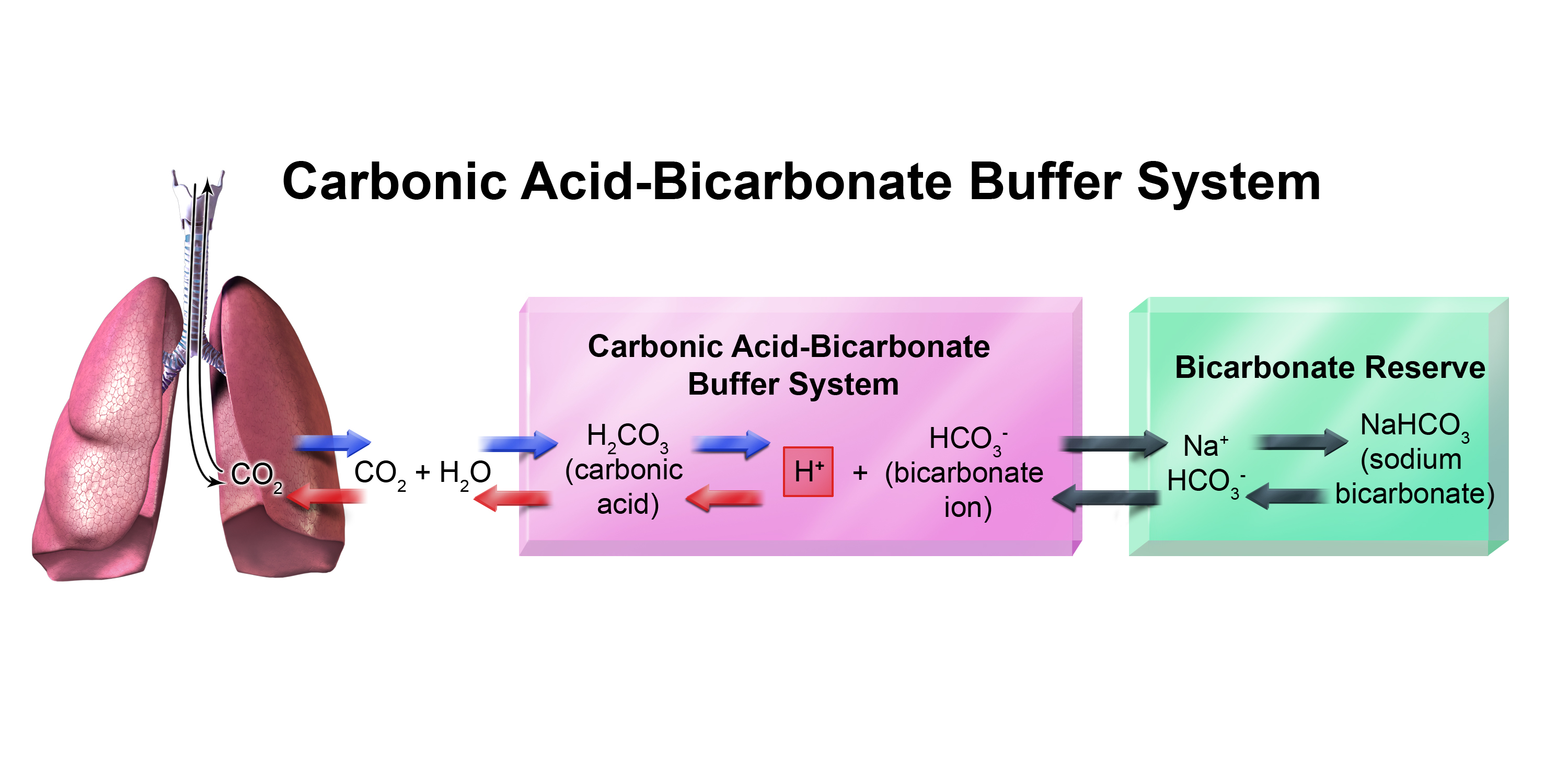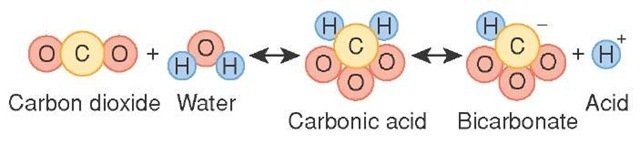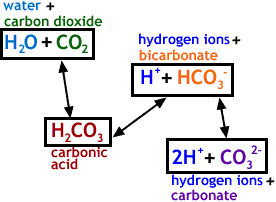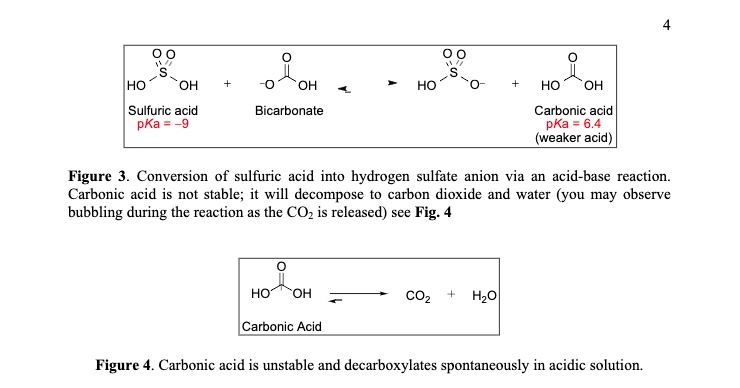
SOLVED: HO OH OH HO HO OH Sulfuric acid pKa Bicarbonate Carbonic acid exd (weaker acid) Figure 3 Conversion of sulfuric acid into hydrogen sulfate anion via an acid-base reaction Carbonic acid
Exogenous citrate can react with carbonic acid to form citric acid and... | Download Scientific Diagram