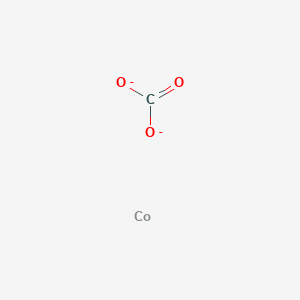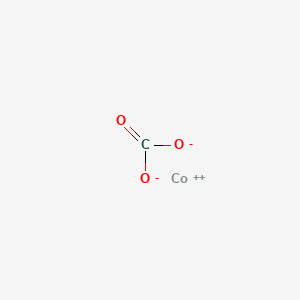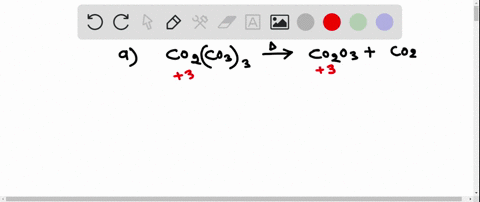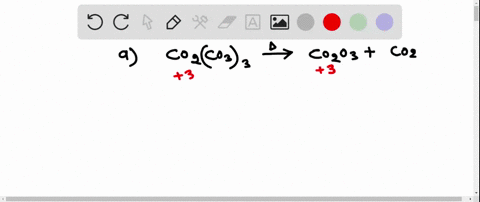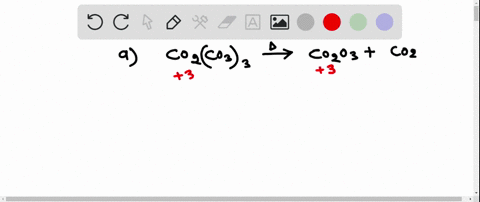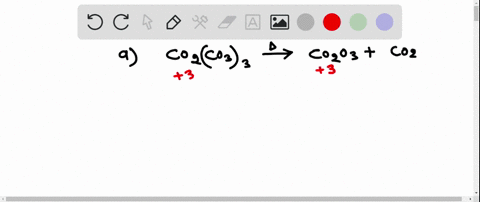
SOLVED:Write a balanced equation for each of the following decomposition reactions: (a) Cobalt(III) carbonate decomposes with heat to give solid cobalt(III) oxide, and carbon dioxide gas. (b) Tin(IV) carbonate decomposes with heat

Q2 Complete the worksheet at your table. Be sure to read instructions carefully and begin working promptly. - ppt download