How is the combustion of carbon a redox reaction? I get the oxidation part, but where is the reduction? - Quora
Combustion always produces carbon dioxide and water”: a discussion of university chemistry students' use of rules in place of principles - Chemistry Education Research and Practice (RSC Publishing) DOI:10.1039/C4RP00089G

The heat of combustion of carbon to CO2 is - 393.5 kJ/mol. The heat released upon formation of 35.2 g of CO2 from carbon and oxygen gas is:
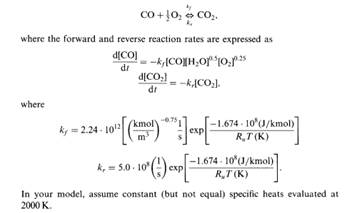
Solved) - Develop a model of the combustion of carbon monoxide with moist... - (3 Answers) | Transtutors
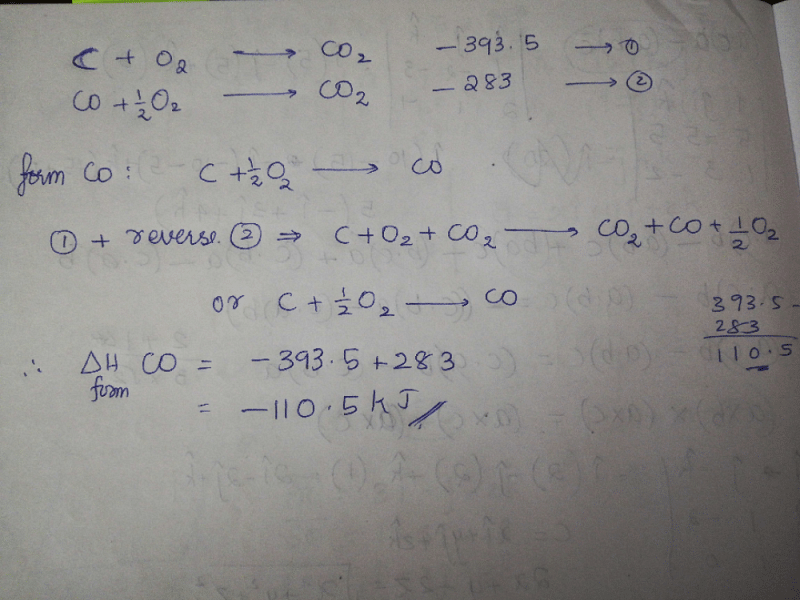
The enthalpies of combustion of carbon and carbon monoxide are -393.5 and -283 KJ mol¹ respectively .The enthalpy of formation of carbon monoxide per mol isa)-110.5 KJb)-676.5 KJc)+676.5 KJd)110.5 KJCorrect answer is

On combustion carbon forms CO and CO2 The heat of formation of CO2 is - 393.5 kJ at of CO is - 110.5 kJ .The heat combustion of CO is
62.The Enthalpies of combustion of carbon and carbon monoxide are 390 kJ and 278kJ respectively. The enthalpy of formation of carbon monooxide is? a) 669 kJ b) 112 kJ c) 112 kJ d) 668 kJ

Ethylene on combustion gives carbon dioxide and water. Its heat of combustion is 1410.0 kJ . mol^-1 . If the heat of formation of CO2 and H2O are 393.3 kJ and 286.2

Enthalpy of combustion of carbon to CO2 is -393.5KJ mol-1. Calculate the heat released upon..... - YouTube

One mole of carbon undergoes incomplete combustion to produce carbon monoxide, Calculate `(Delta H - - YouTube
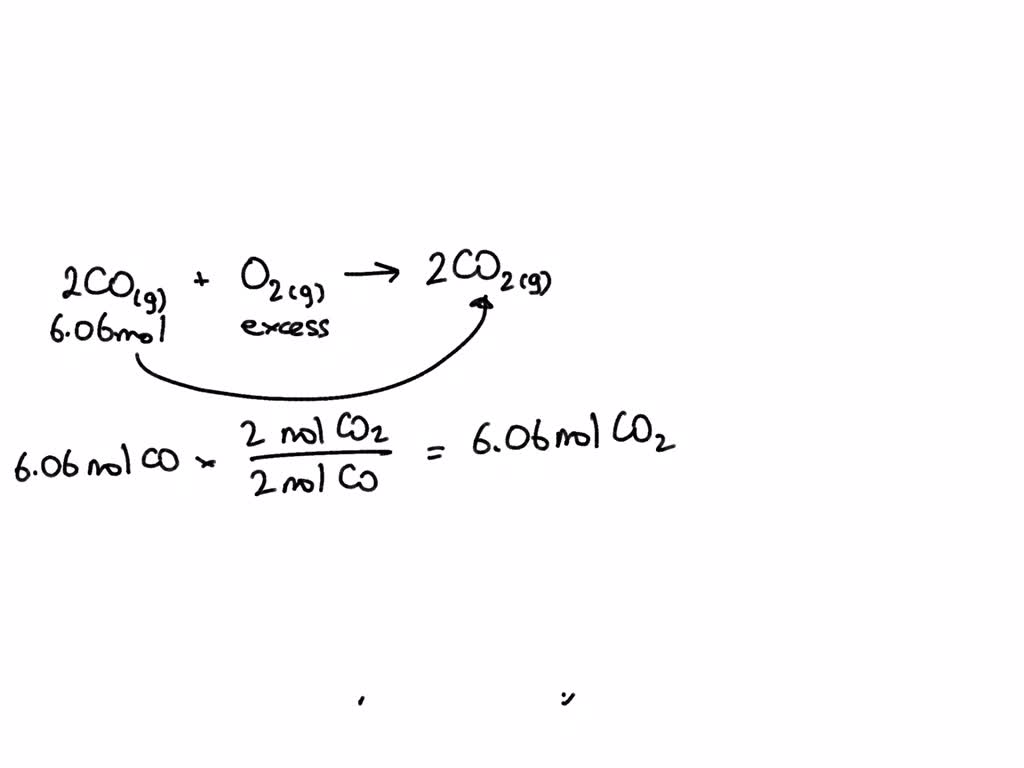
SOLVED: Consider the combustion of carbon monoxide (CO) in oxygen gas: 2CO(g) + O2(g) → 2CO2(g) Calculate the number of moles of CO2 produced if 6.06 moles of CO are reacted with
20. One mole of carbonundergoes incomplete combustion to produce carbon monoxide. Calculate(AH Δ ) for the formation of CO at 298 KGiven R 8314 JK 1 mol 1

Q12 How would you prove experimentally that a Carbon dioxide does not support combustion b Is slight...

Energies | Free Full-Text | A Critical Review of CO2 Capture Technologies and Prospects for Clean Power Generation