62.The Enthalpies of combustion of carbon and carbon monoxide are 390 kJ and 278kJ respectively. The enthalpy of formation of carbon monooxide is? a) 669 kJ b) 112 kJ c) 112 kJ d) 668 kJ

SOLVED: The incomplete combustion of carbon is described by the following equation: 2C () + Oz (g) 2C0 (2) heat energy For this reaction, write the appropriate AH, 4S and AG on
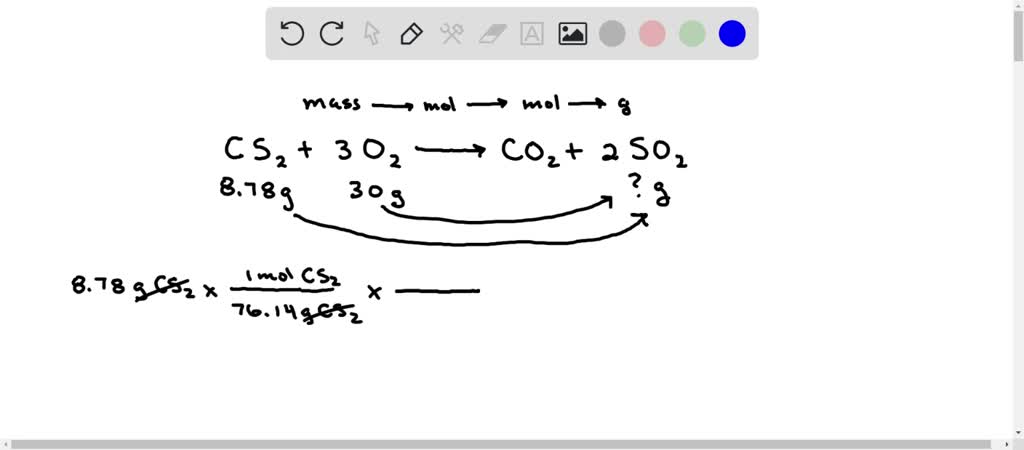
SOLVED: The combustion of carbon disulfide in the presence of excess oxygen yields carbon dioxide and sulfur dioxide according to the following UNBALANCED reaction: CS2 (g) + 3O2 (g) → CO2 (g) +

The enthalpies of combustion of carbon and carbon monoxide are `-390 kJ mol^(-1)` and `-278 kJ mo - YouTube

How is the combustion of carbon a redox reaction? I get the oxidation part, but where is the reduction? - Quora

The heats of combustion of carbon and carbon monoxide are - 393.5 and - 283.5 kJ mol ^-1 , respectively. The heat of formation (in kJ) of carbon monoxide per mole is:

Twitter 上的PhD Natalia Petrovskaya:"Look at the combustion formulas of methane and coal. Carbon dioxide is formed anyway." / Twitter

Write the balanced chemical equation for combustion of carbon dioxide - Science - Chemical Reactions and Equations - 12555075 | Meritnation.com
20. One mole of carbonundergoes incomplete combustion to produce carbon monoxide. Calculate(AH Δ ) for the formation of CO at 298 KGiven R 8314 JK 1 mol 1









![QUESTION 4 [20 POINTS]: The combustion of carbon | Chegg.com QUESTION 4 [20 POINTS]: The combustion of carbon | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F59d%2F59de152d-9b13-4d4d-9997-9e7f3359197d%2FphpYdzZbi.png)