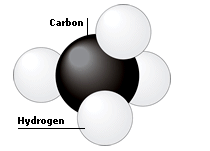
An organic compound contains carbon, hydrogen and oxygen. Its elemental analysis gave C = 38.71% and H, 9.67% and O = 51.62% . The empirical formula of the compound would be:
Investigated molecules, containing carbon double or triple bonds, and... | Download Scientific Diagram

The diagram shows the structures of some substances containing carbon Answer thequestionusing the letters AF Which one of these structures is ionic
35. 12 ml of mixture containing carbon monoxide and nitrogen required 7 ml oxygen to form CO2 and No on combustion the volume of carbon monoxide in the mixture will be

An organic compound contains carbon, hydrogen and oxygen. Its elemental analysis gave`C,38.71% - YouTube
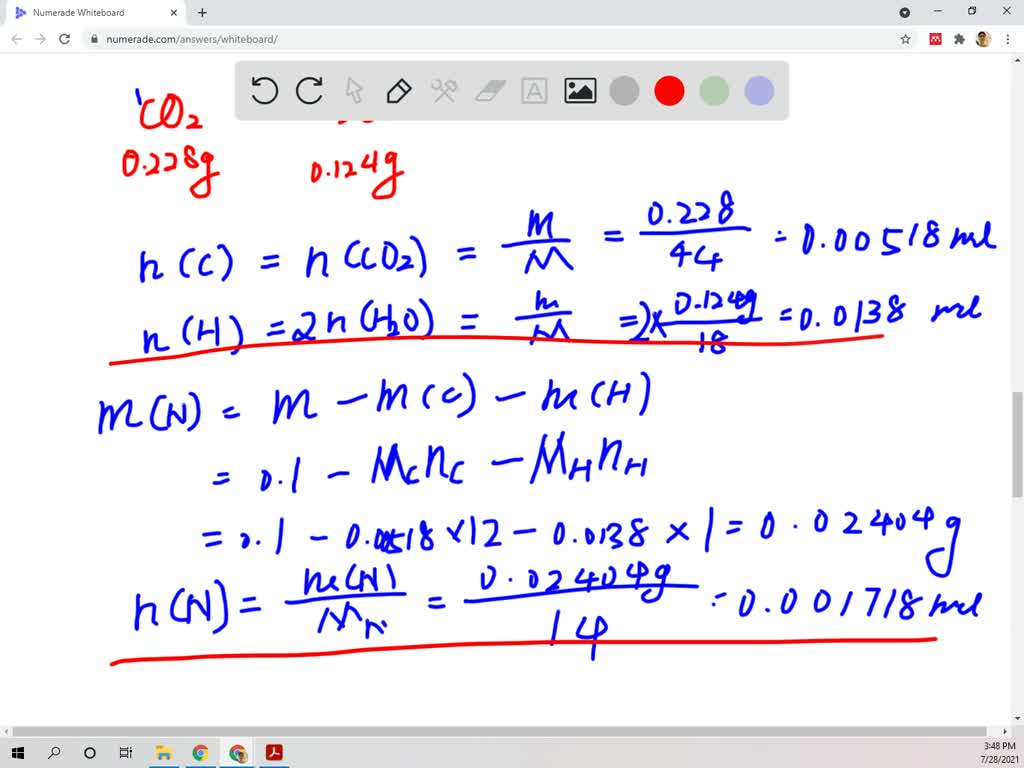
SOLVED: . A compound containing carbon, Hydrogen and Nitrogen only gave the following data on analysis: 0.1g of compound on combustion gave 0.228g of CO2 and 0.124g of H2O. On further analysis,

Molecules | Free Full-Text | A Multifunctional Coating on Sulfur-Containing Carbon-Based Anode for High-Performance Sodium-Ion Batteries

Monodispersed Nitrogen-Containing Carbon Capsules Fabricated from Conjugated Polymer-Coated Particles via Light Irradiation | Langmuir

The empirical formula of an organic compound containing carbon and hydrogen is CH2 . The mass of one litre of N2 is same as one litre of the organic gas. Therefore, the

The structures of six substances containing carbon are shown above.Answer the following questions about these substances Each substance may be used once, more than once or not at all.(a) Which substance, A ,


:max_bytes(150000):strip_icc()/benzene-molecular-model-computer-artwork-487106343-587d46943df78c17b62db25e.jpg)