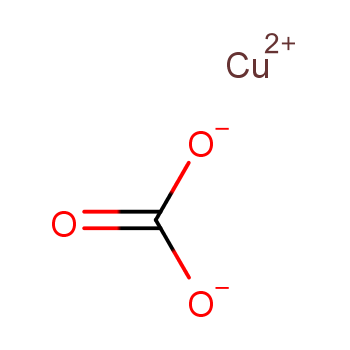
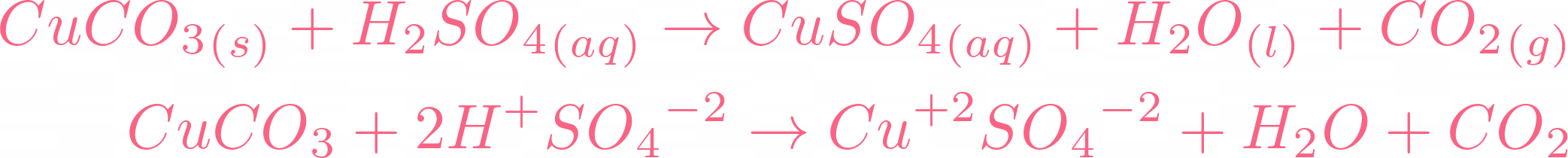SOLVED: Copper carbonate (CuCO3) reacts with hydrochloric acid (HCl) according to this equation: CuCO3(s) + 2HCl(aq) → CuCl2(aq) + H2O(l) + CO2(g). Which statement correctly describes the substances in this reaction? A.

1 st e- transferred 2 nd Ions form 3 rd Opposite ions attract Bond is formed by EMF Compound is made Ionic Compounds. - ppt download

Q3 Give reasons why the following are considered as chemical changes 1 Copper Carbonate on heat give...