Steering carbon dioxide reduction toward C–C coupling using copper electrodes modified with porous molecular films | Nature Communications

Photocatalytic C–C Coupling from Carbon Dioxide Reduction on Copper Oxide with Mixed-Valence Copper(I)/Copper(II) | Journal of the American Chemical Society

Metals. Learning Objectives Order metals in order of reactivity Relate extraction method to reactivity Write word/symbol equations for reduction of metal. - ppt download
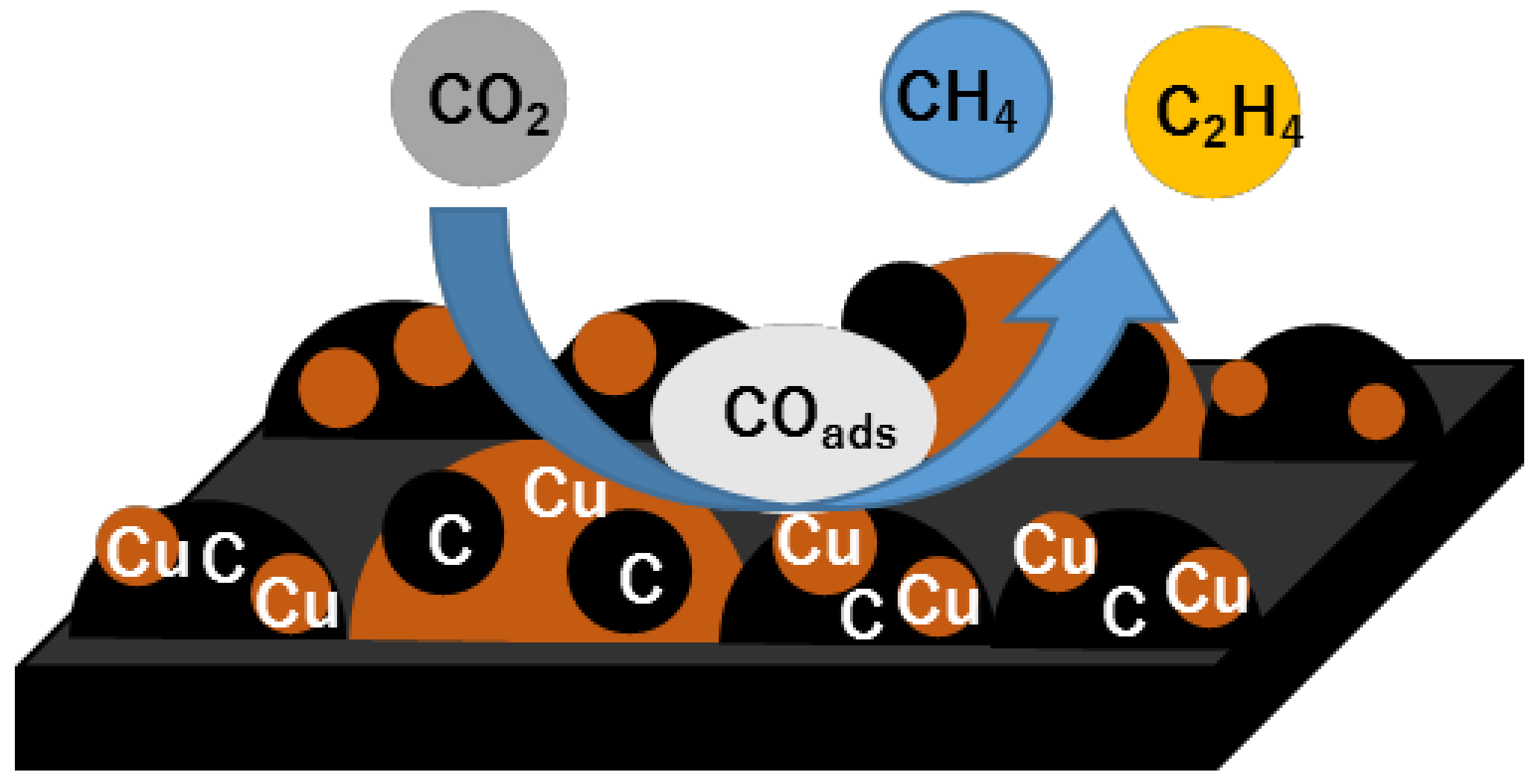
ChemEngineering | Free Full-Text | Electrochemical Carbon Dioxide Reduction in Methanol at Cu and Cu2O-Deposited Carbon Black Electrodes

Gold-in-copper at low *CO coverage enables efficient electromethanation of CO2 | Nature Communications
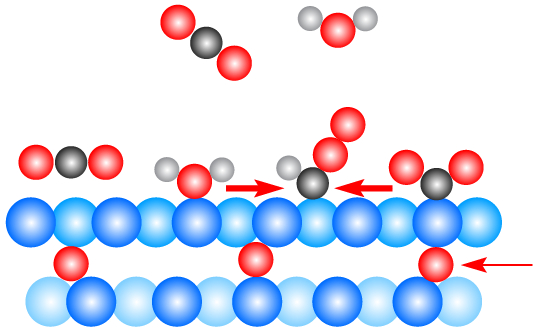
A Surprise Just Beneath Copper Surface in Carbon Dioxide Chemical Reaction | Berkeley Lab – Berkeley Lab News Center
![Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/05/mq2-3.jpg?w=640)
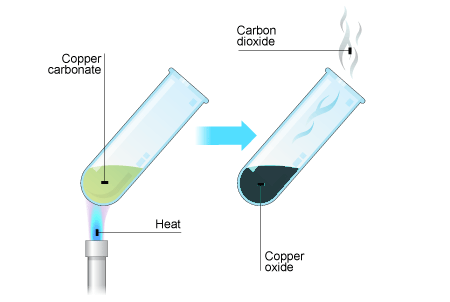
Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition

Electrochemical Carbon Monoxide Reduction on Polycrystalline Copper: Effects of Potential, Pressure, and pH on Selectivity toward Multicarbon and Oxygenated Products | ACS Catalysis
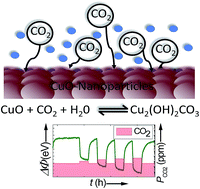
Investigation of CO2 reaction with copper oxide nanoparticles for room temperature gas sensing - Journal of Materials Chemistry A (RSC Publishing)

Scalable Production of Efficient Single-Atom Copper Decorated Carbon Membranes for CO2 Electroreduction to Methanol | Journal of the American Chemical Society