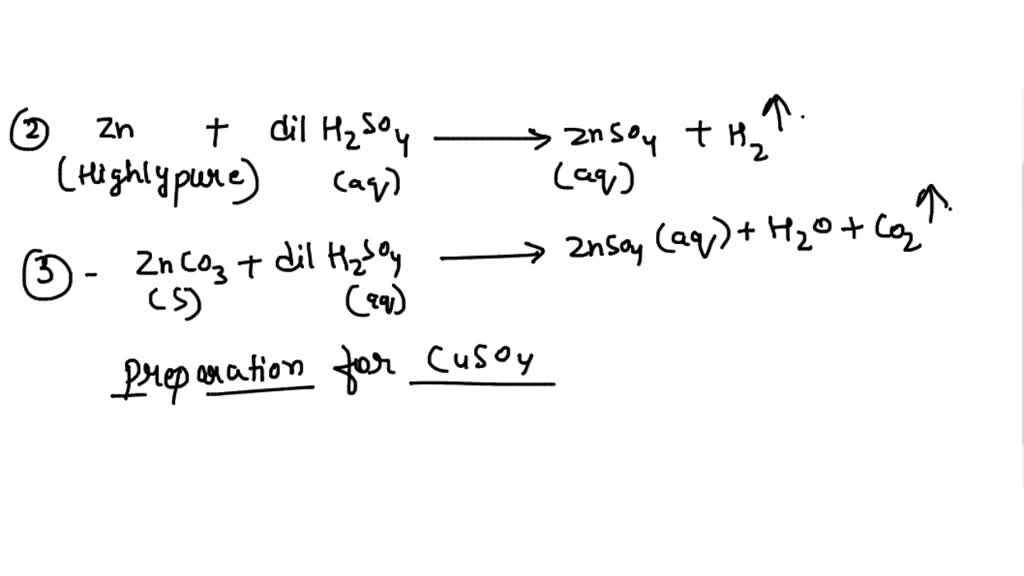
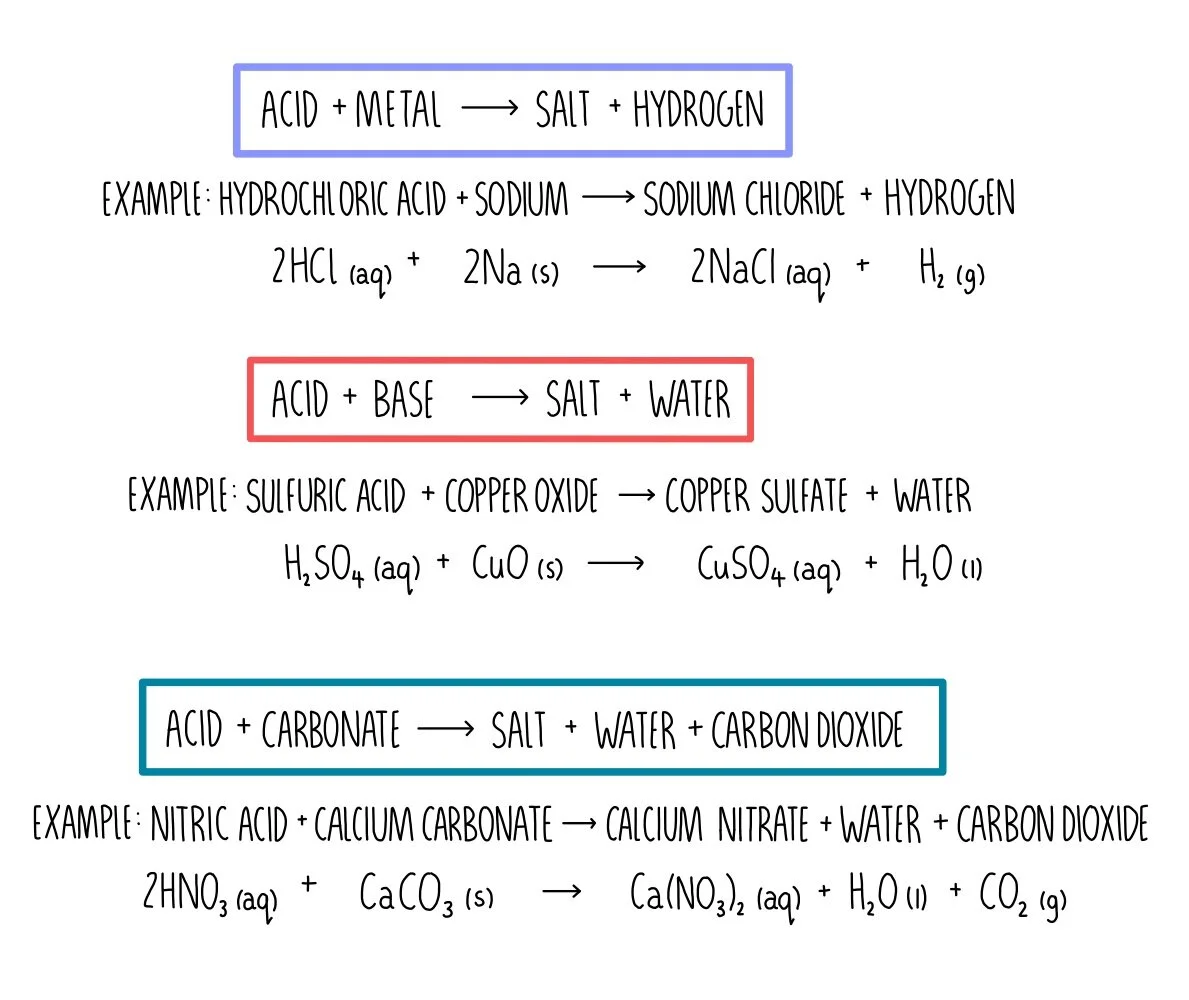
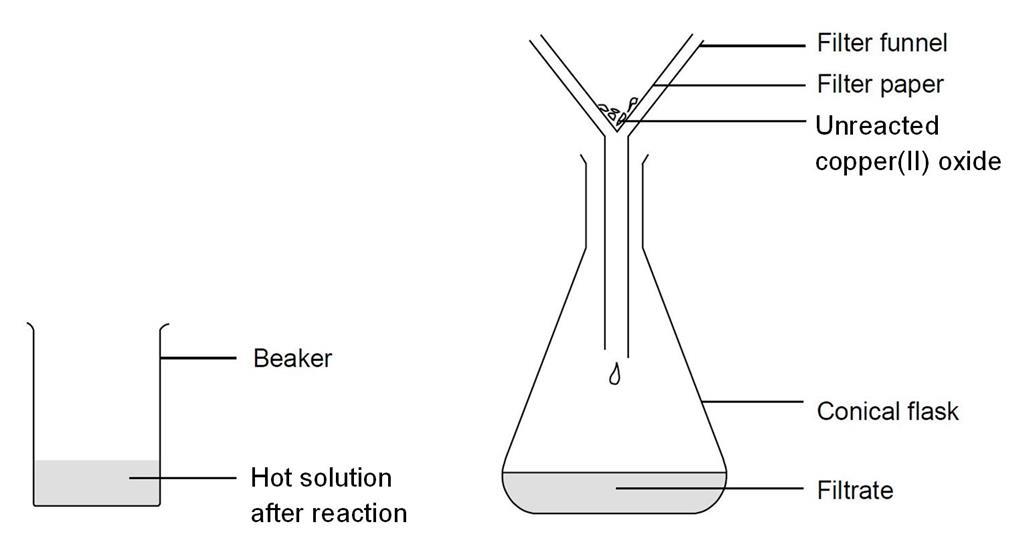
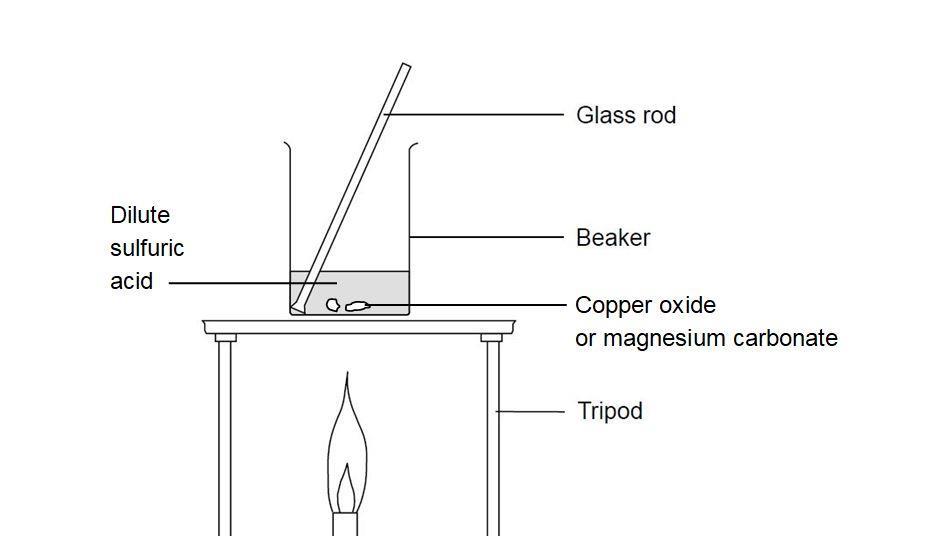
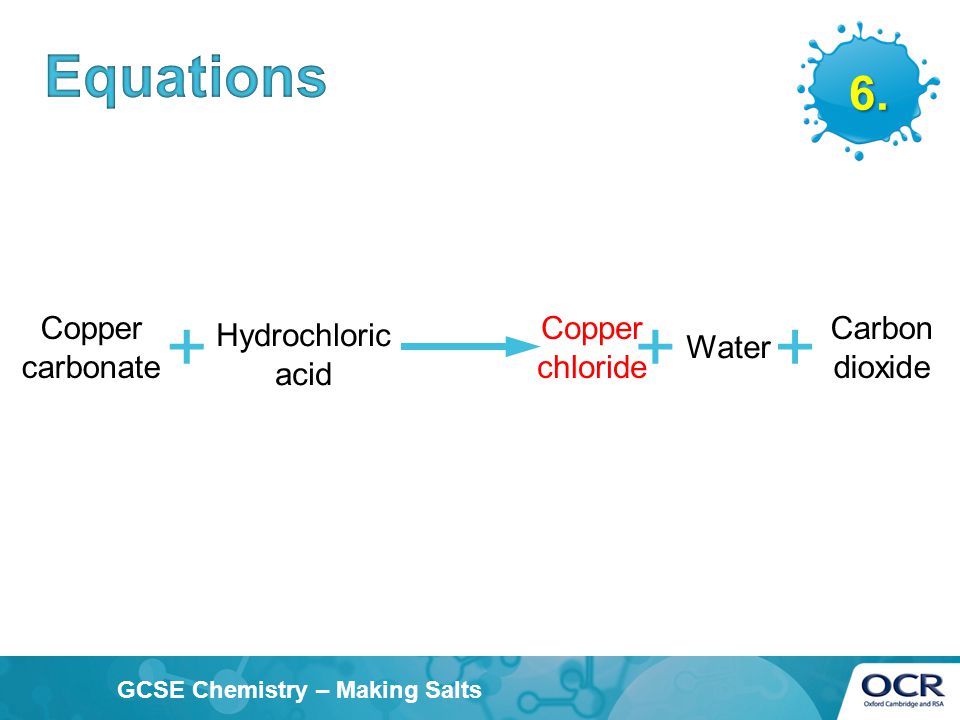
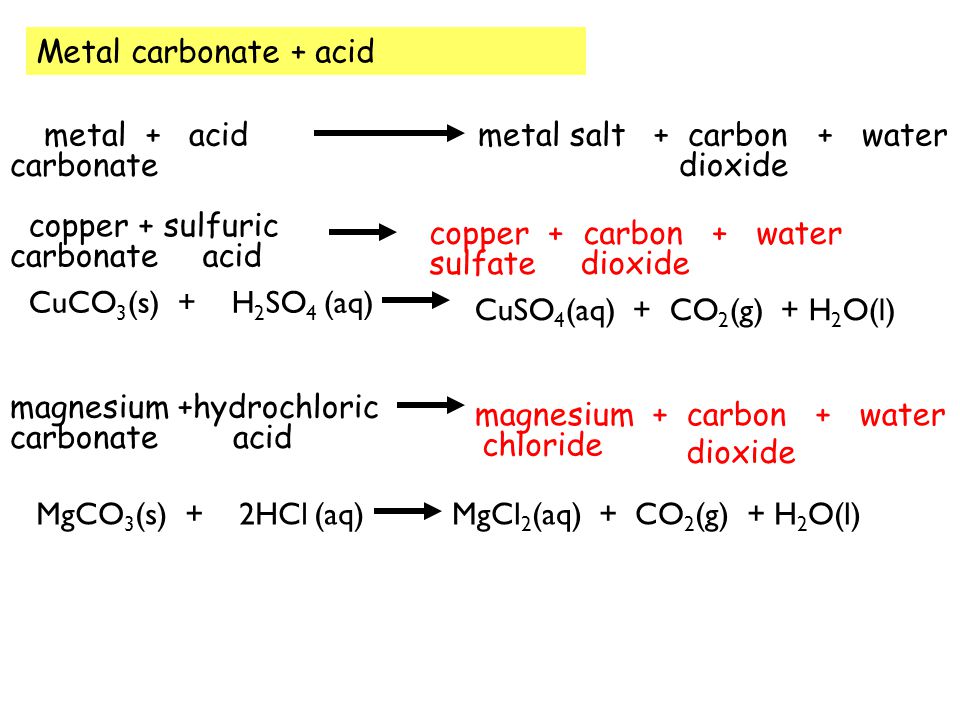
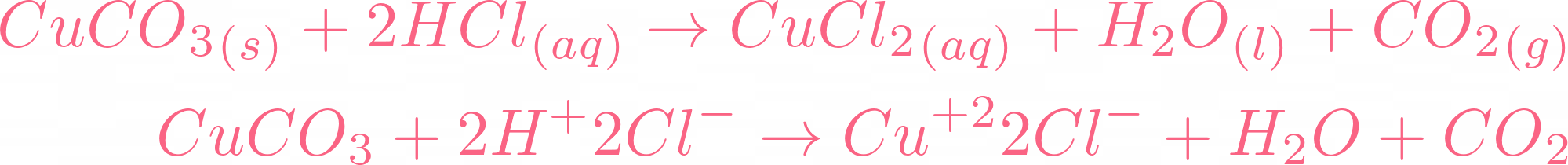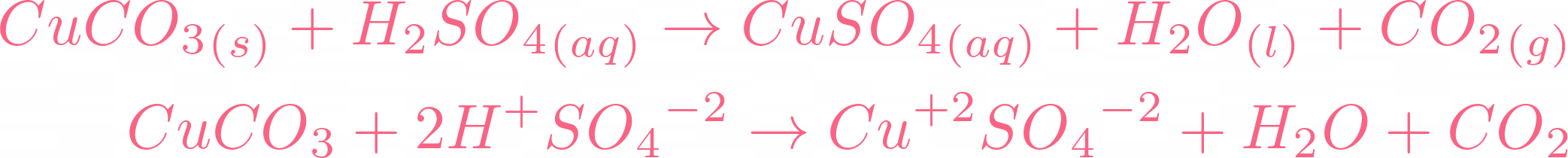How would you write the name of the following compounds ◦ Zn(OH) 2 ◦ NaOH ◦ HCl ◦ Mg(NO 3 ) 2 What does an acid do to red litmus? What does an acid to. - ppt download

Colour changes for simple indicators IndicatorColour in acidColour in alkali litmusredblue methyl orangeredyellow phenolphthaleincolourlessred. - ppt download
![Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/05/mq2-3.jpg?w=640)
Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition
i) Lead sulphate from lead carbonate (ii) Sodium sulphate using dilute sulphuric acid. (iii) Copper chloride using copper carbonate. - Sarthaks eConnect | Largest Online Education Community