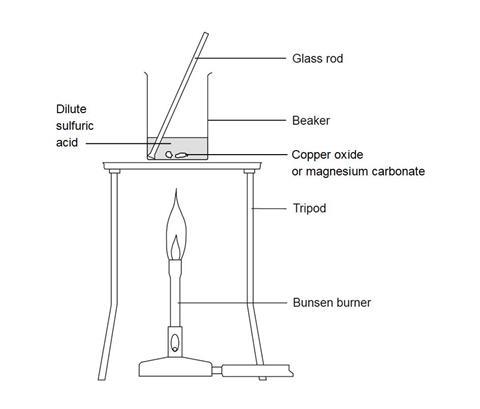
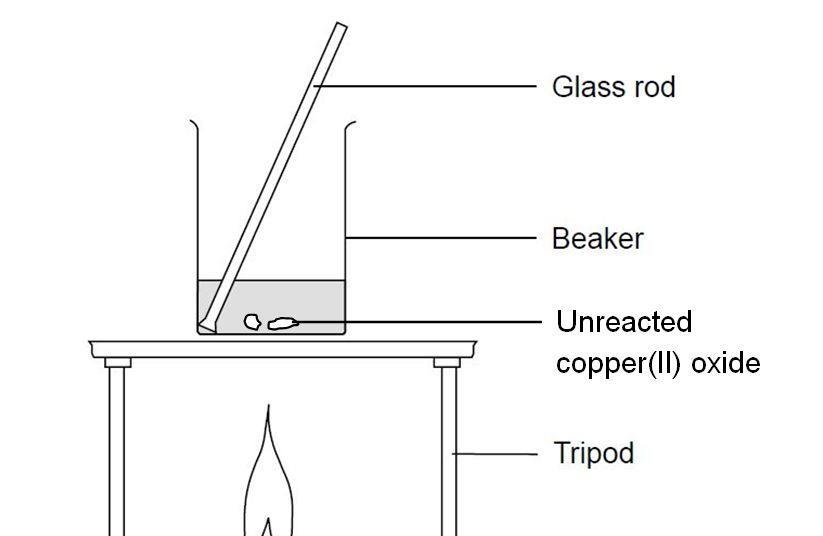
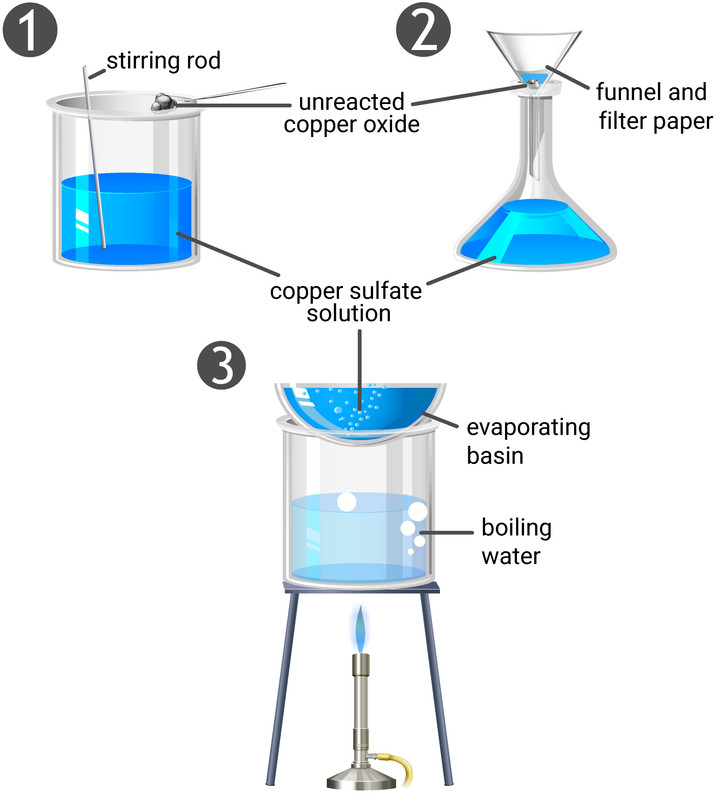
Practical: Prepare Copper(II)Sulfate (2.7.8) | Edexcel IGCSE Chemistry Revision Notes 2019 | Save My Exams

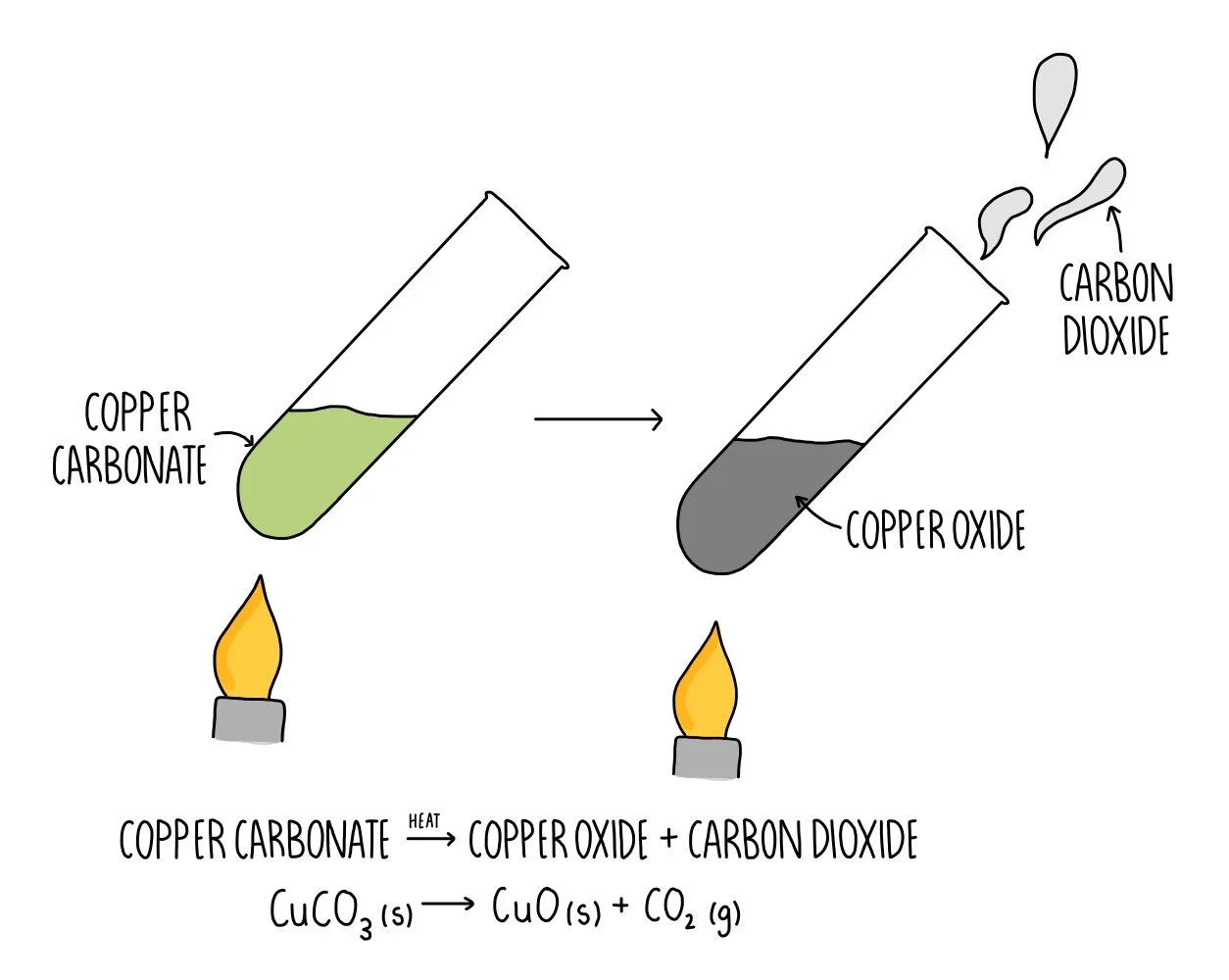
Proust took two samples of copper carbonate a compound of copper carbon and oxygen He took a sample from nature and another sample prepared in the lab and decomposed it chemically to
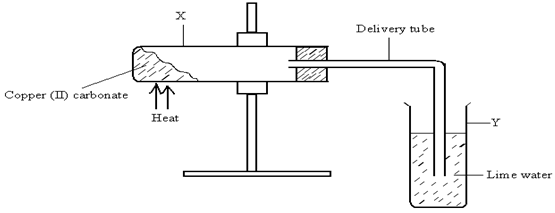
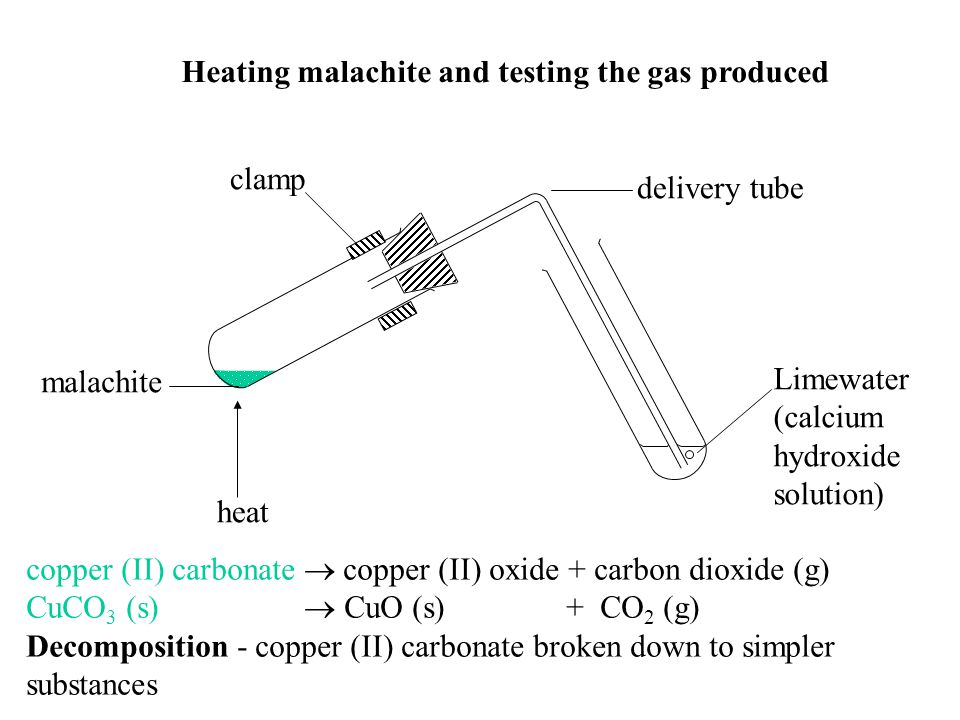
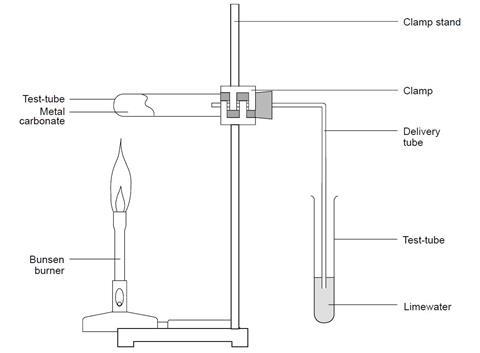
The diagram below illustrates an experiment set up to investigate the effect of heat on copper (II) carbonate. Study it and answer the questions that follow. - EasyElimu Questions and Answers

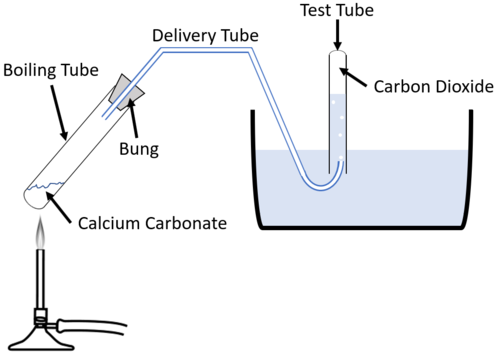
P A student carried out an experiment to investigate the rate of reaction between powdered calcium carbonate and excess dilute hydrochloric acid, using the following apparatus. The student measured the mass of