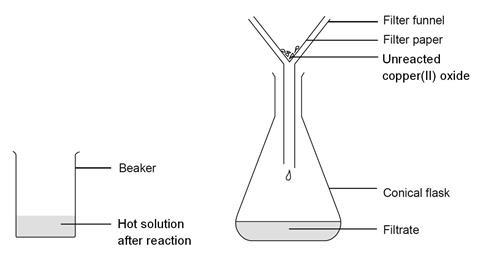
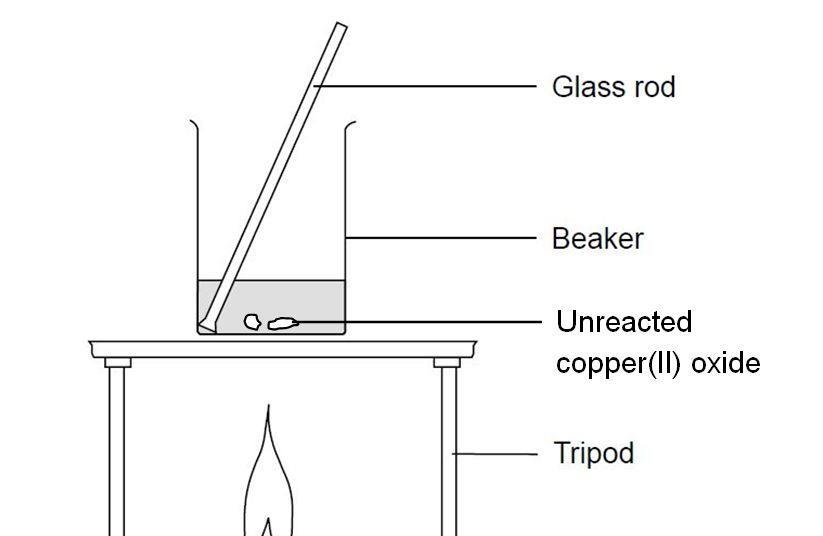
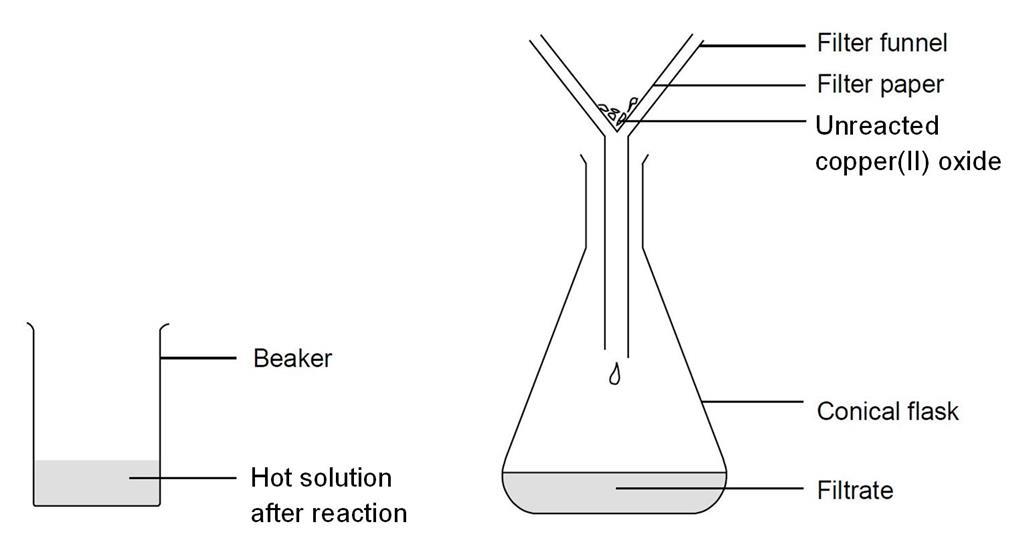
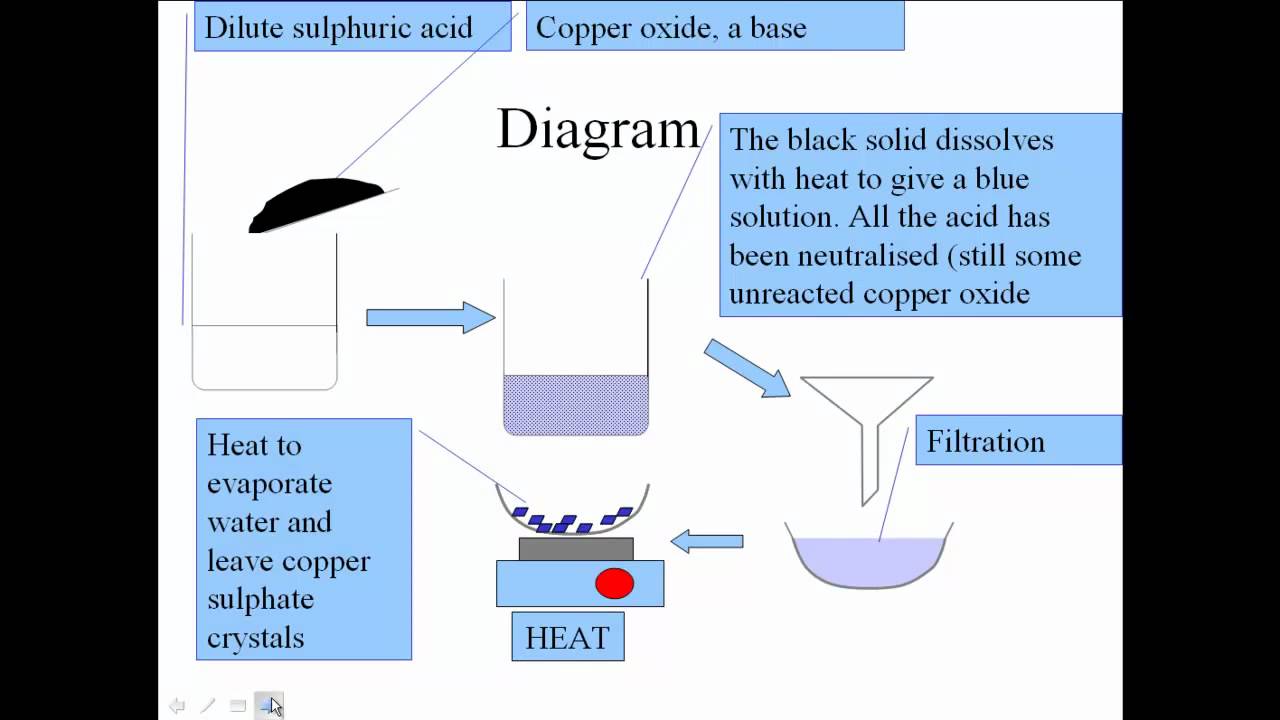
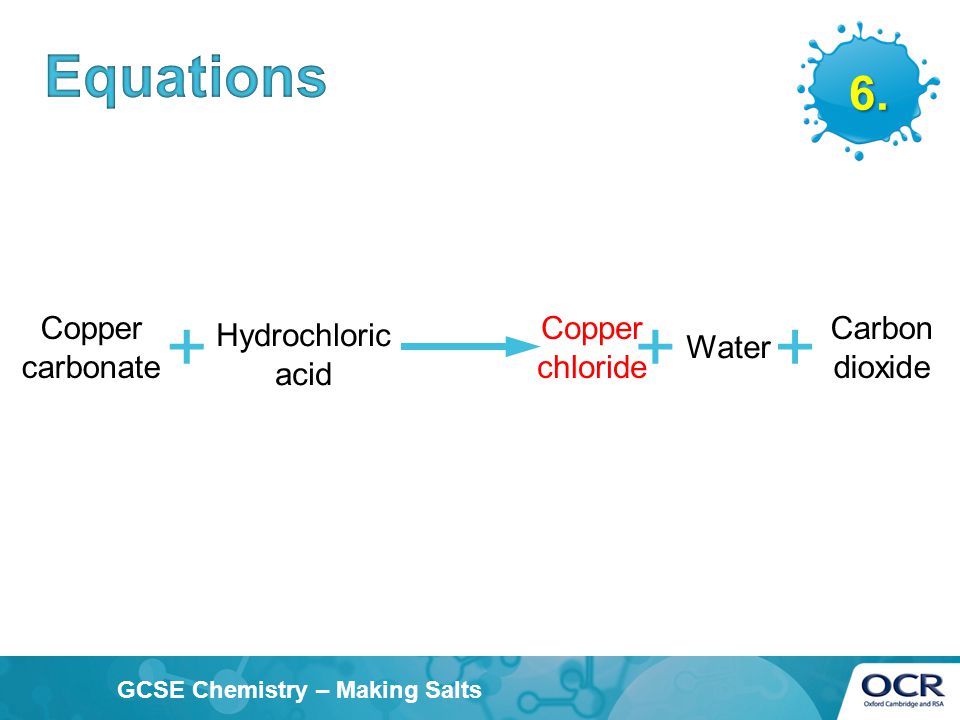
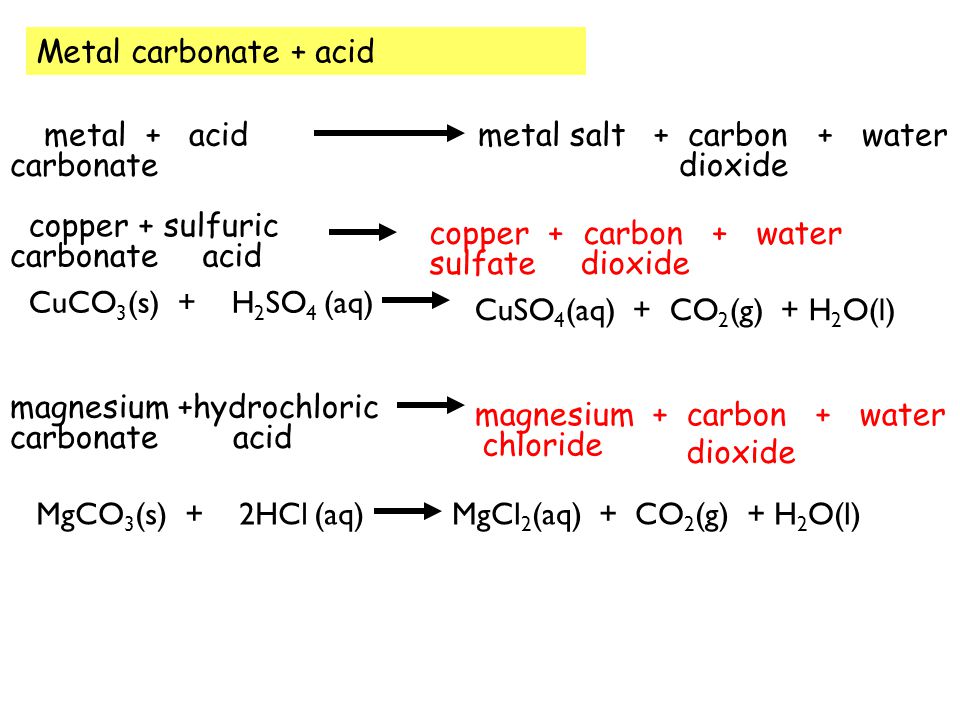
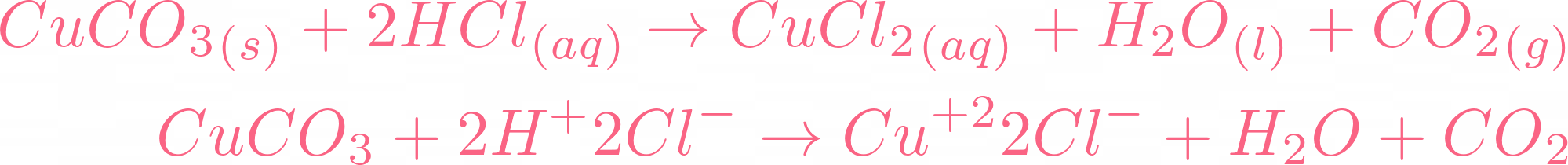Practical - preparation of crystals of a soluble salt - Making salts - Eduqas - GCSE Combined Science Revision - Eduqas - BBC Bitesize

H 2 SO 4 + Zn 1) Sulphuric acid + zinc 3) Nitric acid + sodium thiosulphate 2) Hydrochloric acid + magnesium 4) Hydrogen peroxide with catalyst Popular. - ppt download

![AUFBAU1 [METALS: COPPER COMPOUNDS (2)] AUFBAU1 [METALS: COPPER COMPOUNDS (2)]](https://www.wissensdrang.com/media/scheme2.gif)