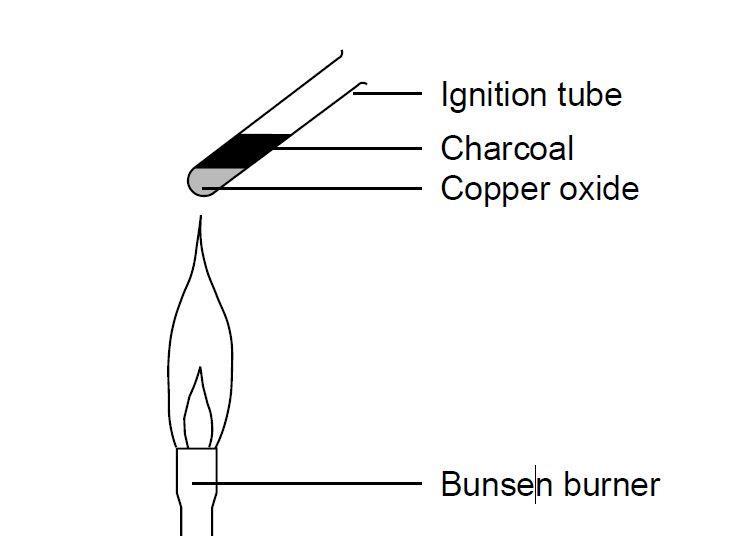
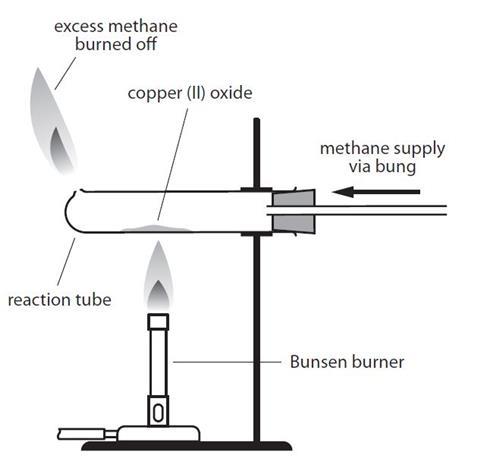
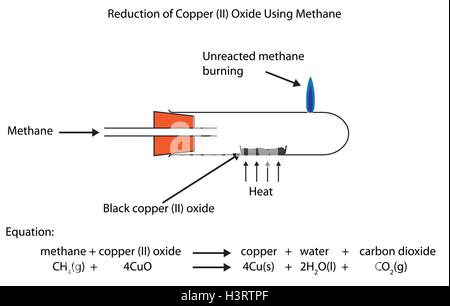
Metals. Learning Objectives Order metals in order of reactivity Relate extraction method to reactivity Write word/symbol equations for reduction of metal. - ppt download

Write balanced chemical equations for the following word equation:Copper + Oxygen → Copper (II) oxide
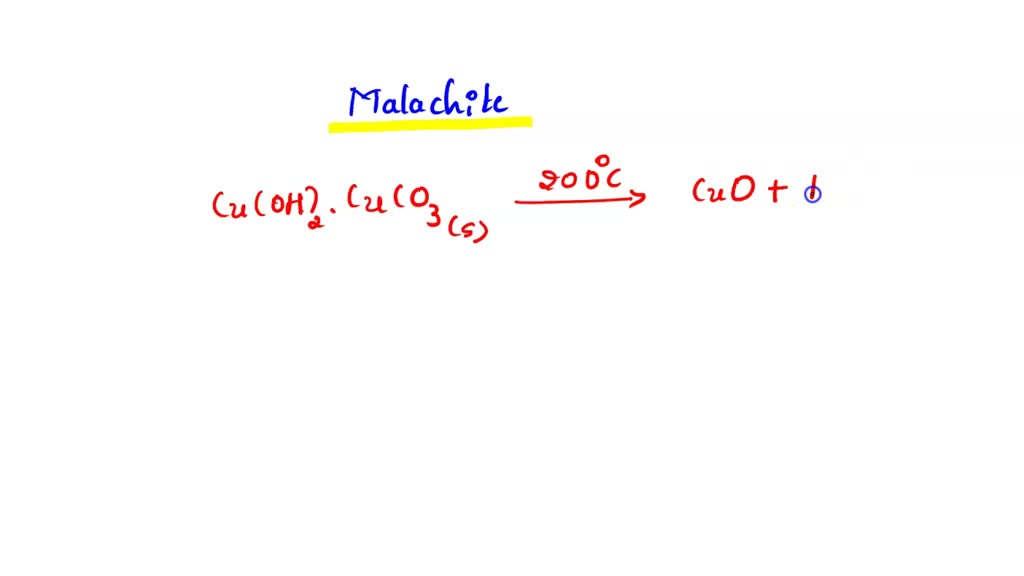
SOLVED: When malachite is decomposed, copper(II) oxide, carbon dioxide and water vapor are formed. Write the balanced chemical equation.

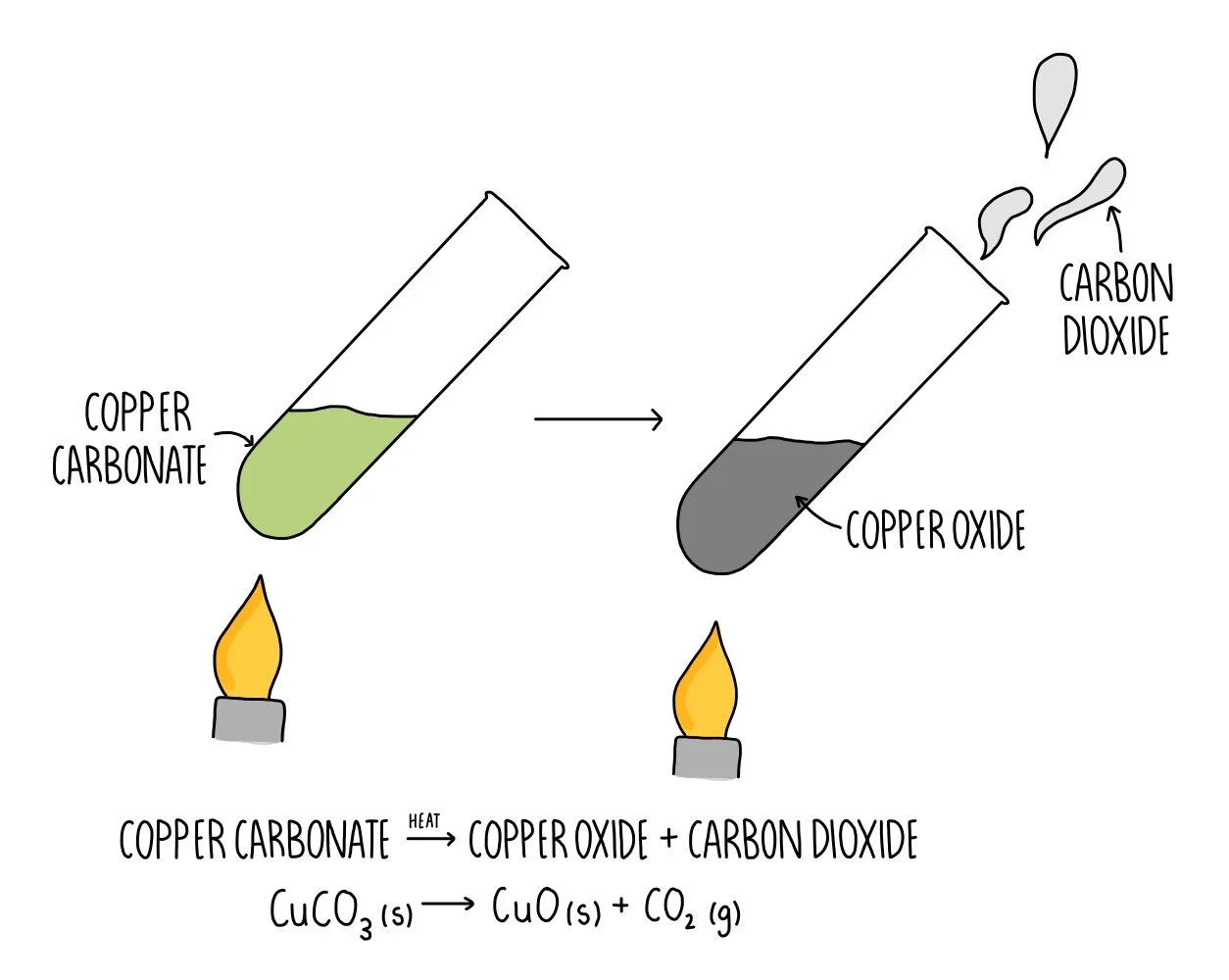
Water gas3Copper oxide and carbon dioxide are made when copper carbonate is heated strongly. Whichis a - Brainly.com

Q3 Give reasons why the following are considered as chemical changes 1 Copper Carbonate on heat give...

Nano- and Microstructured Copper/Copper Oxide Composites on Laser-Induced Carbon for Enzyme-Free Glucose Sensors | ACS Applied Nano Materials
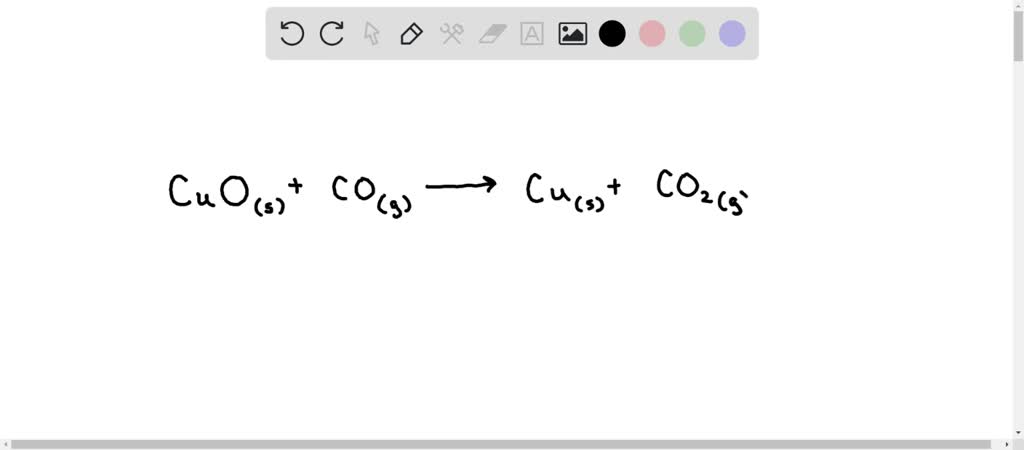
SOLVED: Enter a balanced equation for the reaction between solid copper(II) oxide and carbon monoxide gas that produces solid copper and carbon dioxide gas. Express your answer as a chemical equation. Identify

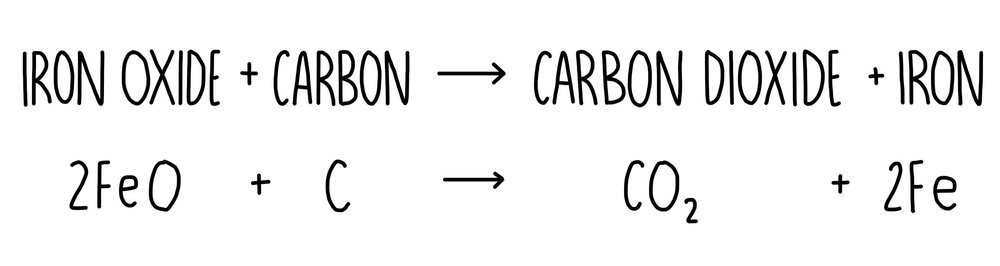
Extracting Copper Learning Objective Describe the extraction of copper. All Most Some state that copper is mined in the form of its ores. (D) describe. - ppt download