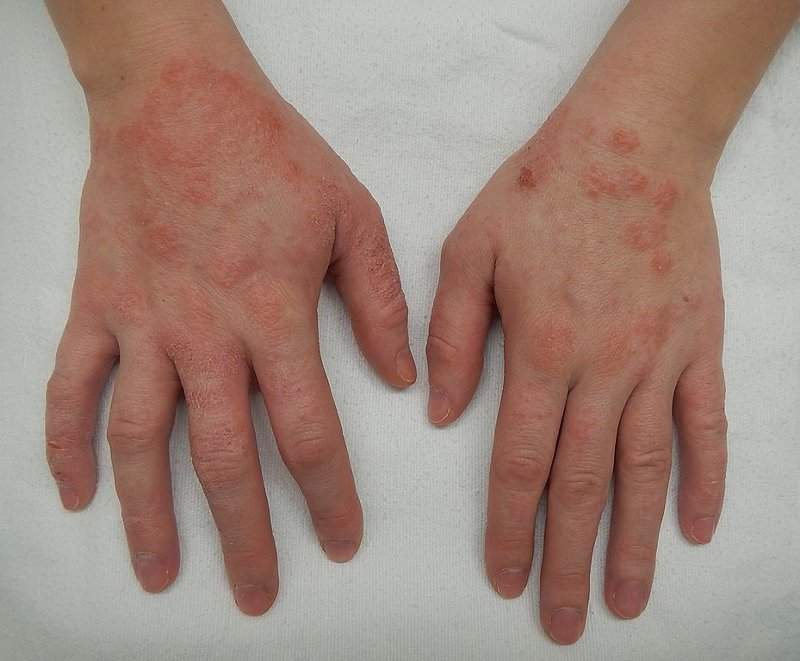
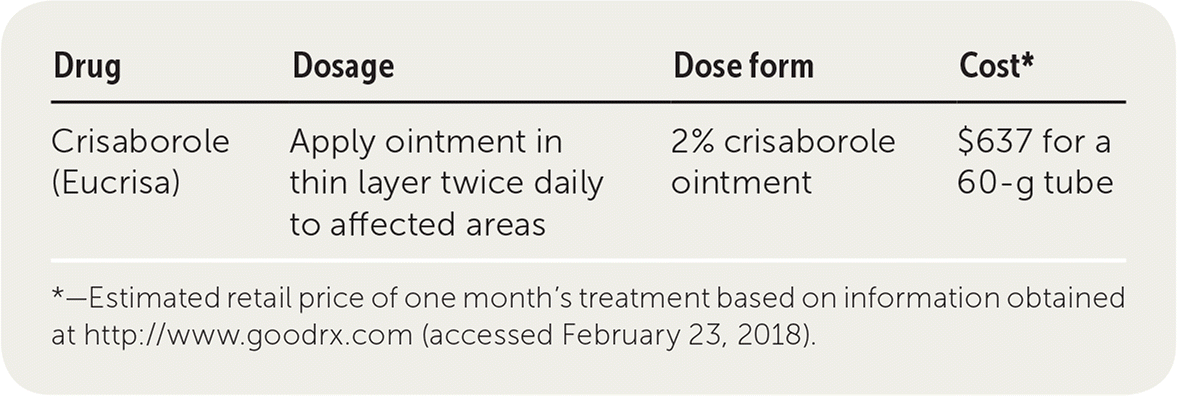Eczema Support Australia - PBAC MEETING – Call for comments – DEADLINE 7 October: The era of eczema is upon us… this means that, finally, new and innovative treatment options are becoming

Use of Topical Crisaborole for Treating Dermatitis in a Variety of Dermatology Settings Topical Crisaborole for Treating Dermatitis in Dermatology

Positive Top-Line Results Seen in Study of Crisaborole for Mild to Moderate AD in Young Children - Practical Dermatology

Crisaborole Topical Ointment, 2%: A Nonsteroidal, Topical, Anti-Inflammatory Phosphodiesterase 4 Inhibitor in Clinical Development for the Treatment of Atopic Dermatitis - JDDonline - Journal of Drugs in Dermatology
![PDF] Topical Treatment Utilization for Patients with Atopic Dermatitis in the United States and Budget Impact Analysis of Crisaborole Ointment, 2% by Ryan Clark, Duygu Bozkaya, Mark Levenberg, Steven Faulkner, Timothy W. PDF] Topical Treatment Utilization for Patients with Atopic Dermatitis in the United States and Budget Impact Analysis of Crisaborole Ointment, 2% by Ryan Clark, Duygu Bozkaya, Mark Levenberg, Steven Faulkner, Timothy W.](https://og.oa.mg/Topical%20Treatment%20Utilization%20for%20Patients%20with%20Atopic%20Dermatitis%20in%20the%20United%20States%20and%20Budget%20Impact%20Analysis%20of%20Crisaborole%20Ointment%2C%202.png?author=%20Ryan%20Clark,%20Duygu%20Bozkaya,%20Mark%20Levenberg,%20Steven%20Faulkner,%20Timothy%20W.%20Smith,%20Robert%20A.%20Gerber)
PDF] Topical Treatment Utilization for Patients with Atopic Dermatitis in the United States and Budget Impact Analysis of Crisaborole Ointment, 2% by Ryan Clark, Duygu Bozkaya, Mark Levenberg, Steven Faulkner, Timothy W.