The refractive index of carbon tetrachloride is 1.4575 and its density is 1.598 x 10 ^ 3 kg m ^ -3. - Brainly.in

A container is partly filled with carbon tetrachloride (CCl4) of density 1.6 × 10^3 kg/m^3 . Water (which does not mix with CCl4 ) is poured on top. A block of plastic
Answer on Question 52989, Physics, Mechanics | Kinematics | Dynamics Question: A reservoir of carbon tetrachloride (����
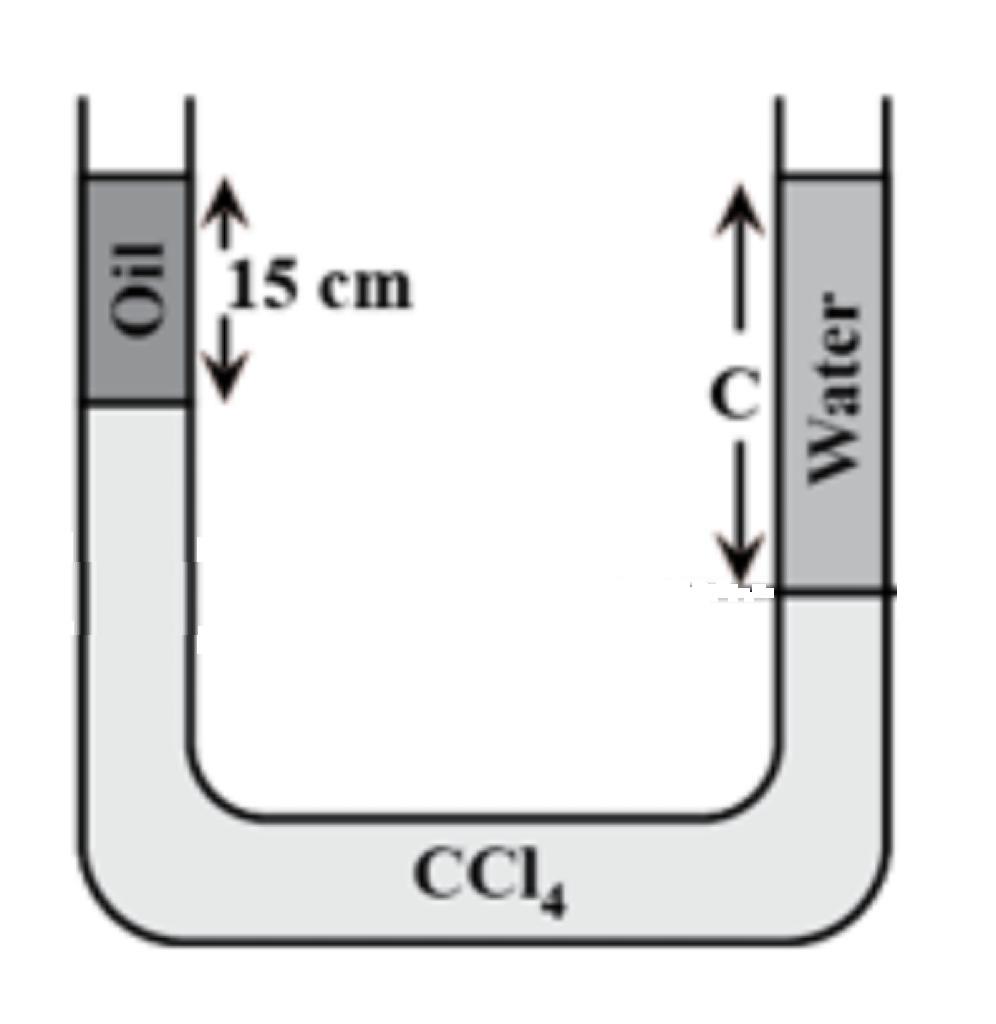
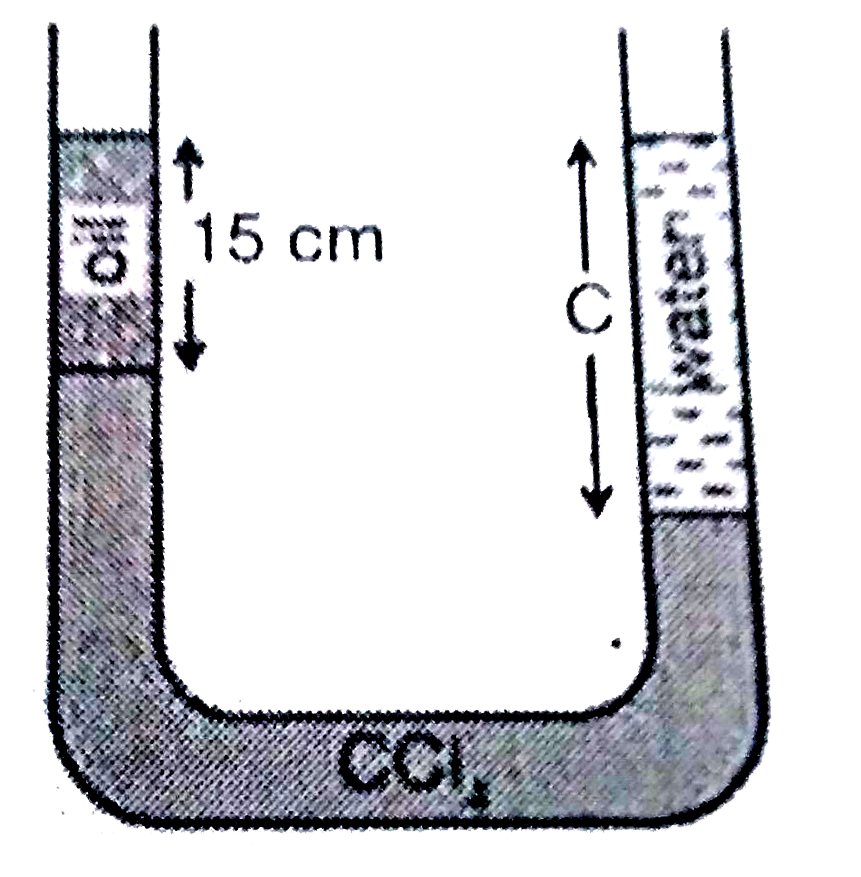
A U tube contains oil carbon tetrachloride and water as shown in the figure The density of oil is08gcm3and that of carbon tetrachloride is16gcm3 If oil and watersurfacesare at the same level
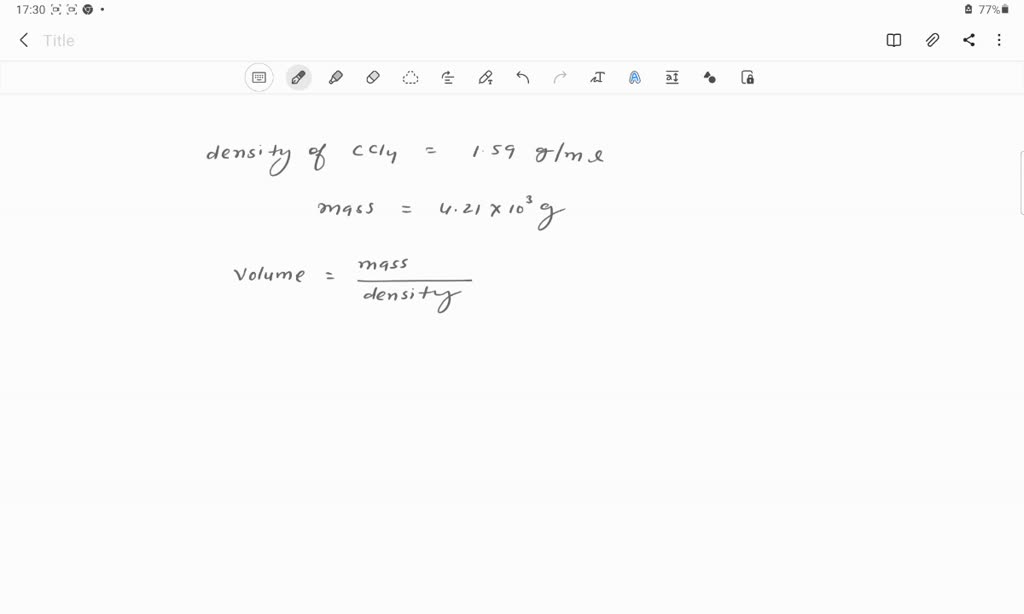
SOLVED: if the density of carbon tetrachloride is 1.59g/mL, what is the volume in LITERS of 4.21 x 10^3 grams of carbon tetrachloride?

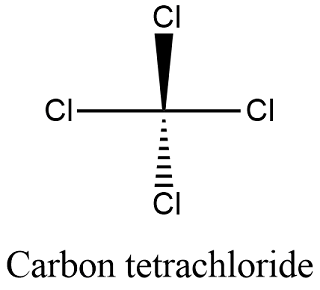
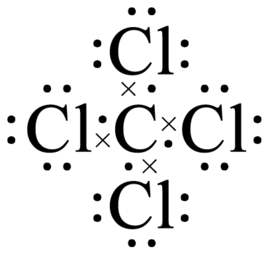
Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar
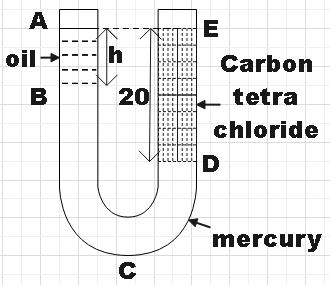
Calculate h in the U-tube shown in the figure. (Density of oil =$0.9gc{{m}^{-3}}$, density of carbon tetrachloride = $1.6gc{{m}^{-3}}$ and density of mercury =$13.6gc{{m}^{-3}}$). \n \n \n \n \n