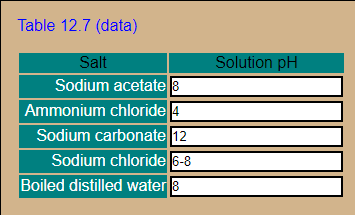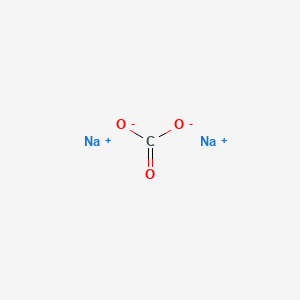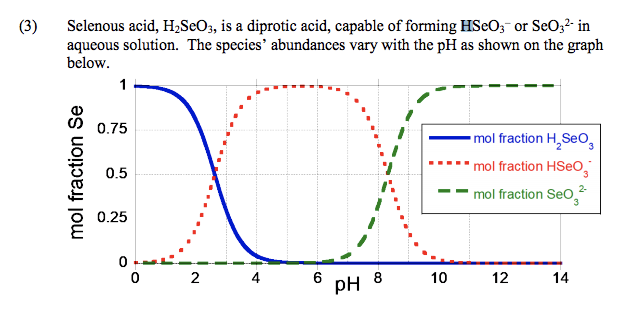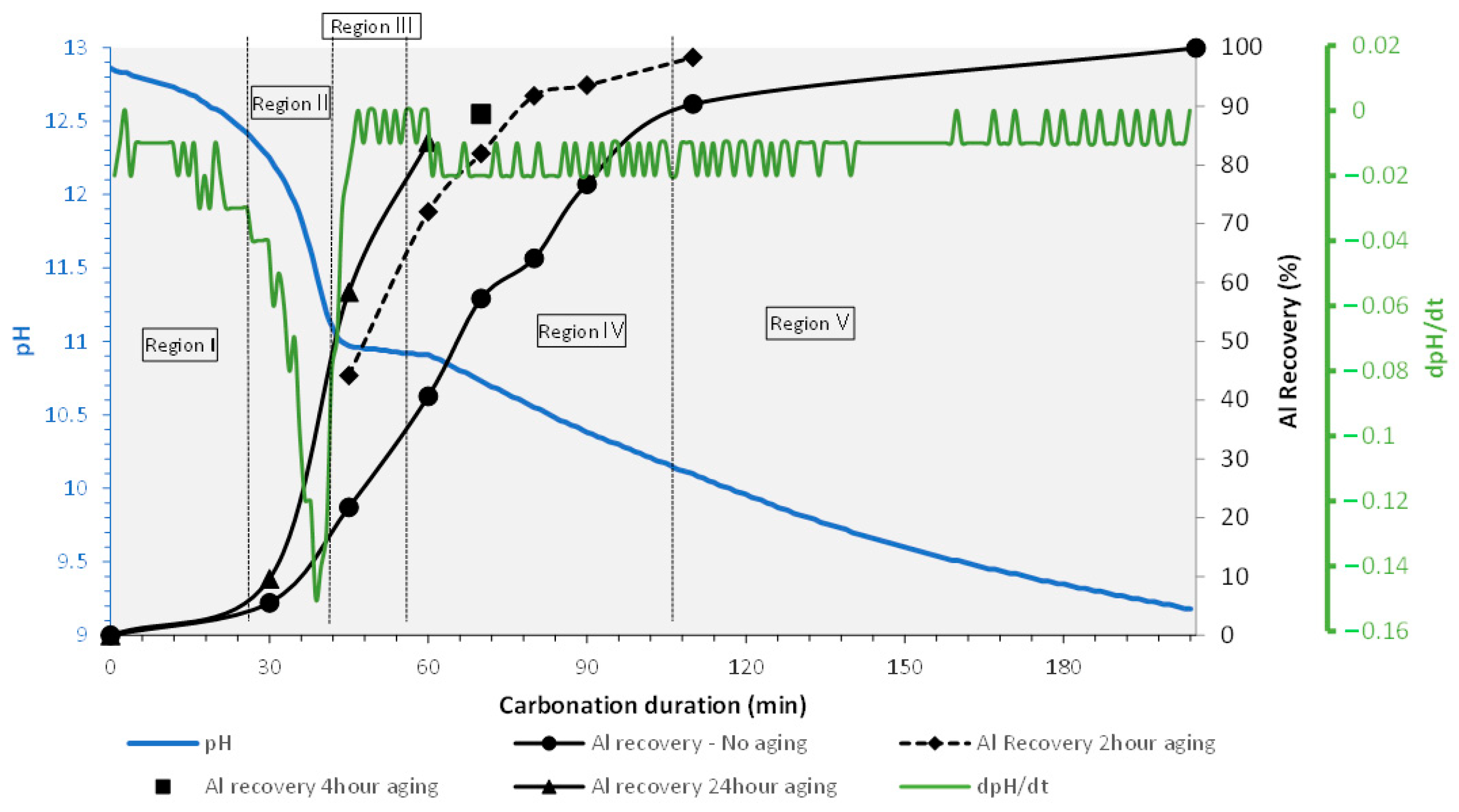
Crystals | Free Full-Text | Carbonation of Sodium Aluminate/Sodium Carbonate Solutions for Precipitation of Alumina Hydrates—Avoiding Dawsonite Formation
![PDF] The pH Monitoring of the non-steady CO 2 Absorption Process in Aqueous Sodium Carbonate Solutions | Semantic Scholar PDF] The pH Monitoring of the non-steady CO 2 Absorption Process in Aqueous Sodium Carbonate Solutions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6065d17c8110019cced7e758e71c3999ee1df481/4-Table3-1.png)
PDF] The pH Monitoring of the non-steady CO 2 Absorption Process in Aqueous Sodium Carbonate Solutions | Semantic Scholar

Waste gypsum conversion and pH for different sodium carbonate/gypsum... | Download Scientific Diagram

Influence of pH at 0.6 M Na ? (a) and of sodium carbonate at pH 10 (b)... | Download Scientific Diagram

SOLVED: Calculate the pH of a 0.1 M solution of sodium carbonate (𝑁𝑎2𝐶𝑂3), given the following information. 𝐾𝑏1 =2.1×10−4, 𝐾𝑏2 =2.2×10−8 Species of interest: 𝐶𝑂32−, 𝐻𝐶𝑂3−, 𝐻2𝐶𝑂3

Influence of pH at 0.6 M Na + (a) and sodium carbonate at pH 10 (b) on... | Download Scientific Diagram




![PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aaa98c1f9de4c398b6a057bf4936ea76736034c7/1-Table1-1.png)