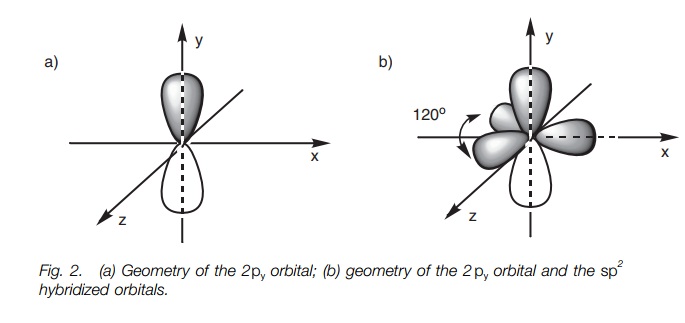
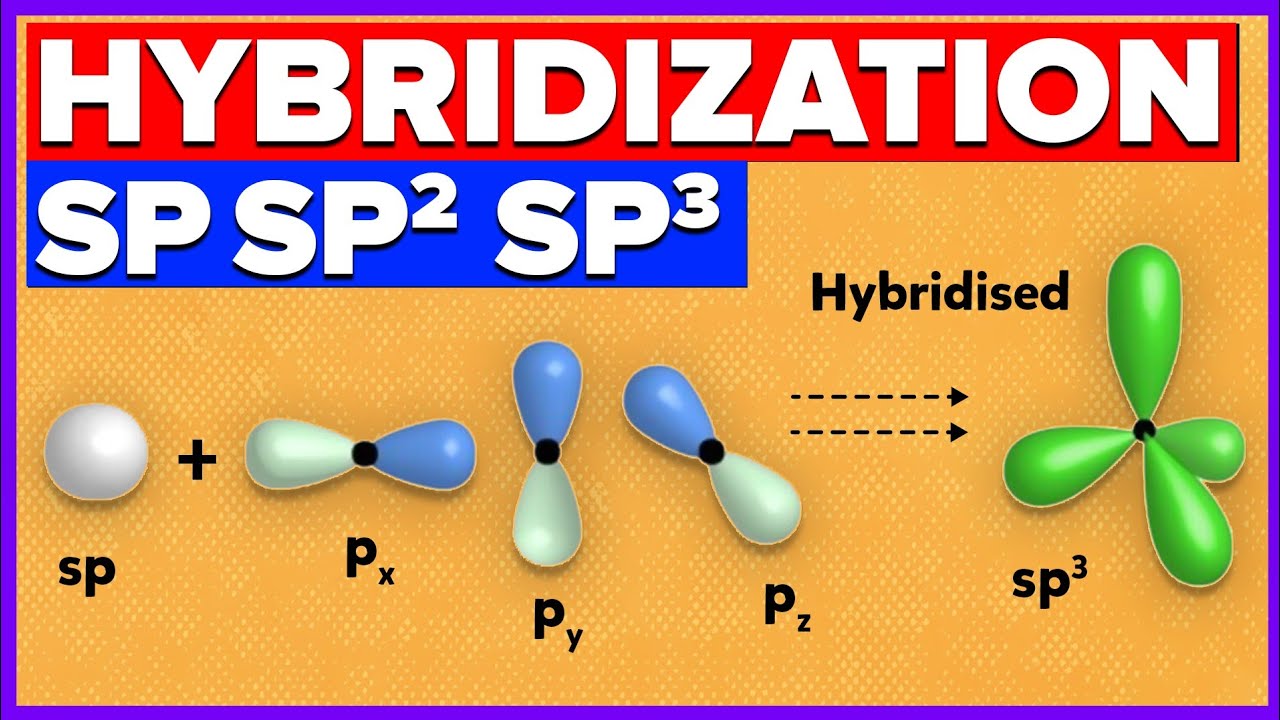
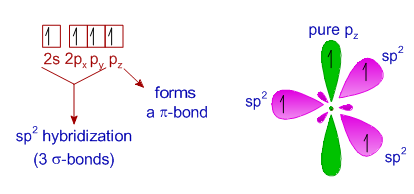
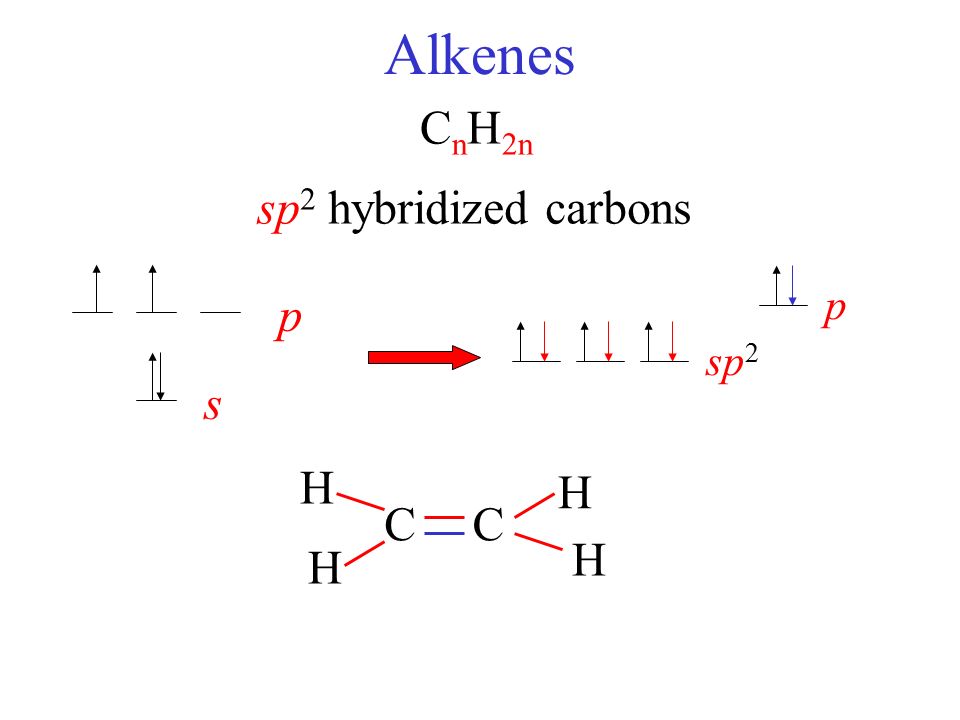
Why does carbon have different hybridization? Sometimes it shows sp3, but sometimes it show sp2. Why is the one p orbital left unhybridized? Does it depend on some situation or something else? -

A structure for a molecule that meet the following description: Contains two sp2-hybridized carbons and two sp3-hybridized carbons | Homework.Study.com
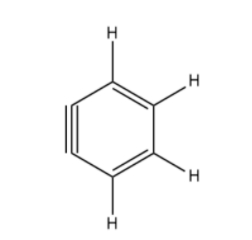
How many $s{{p}^{2}}$ and sp- hybridized carbon atoms are present respectively in the following compound?\n \n \n \n \n (A) 4,2(B) 6,0(C) 3,3(D) 5,1
What is the difference between sp3 hybridization and sp2 hybridization for carbon atoms? Which one is more stable and why? - Quora


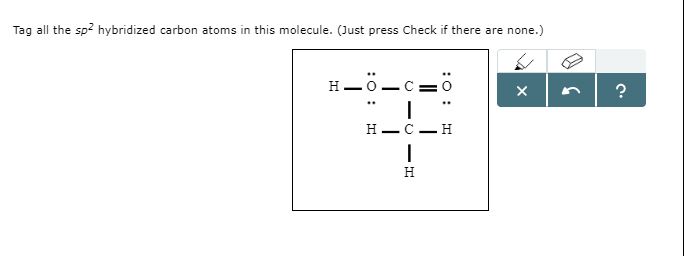


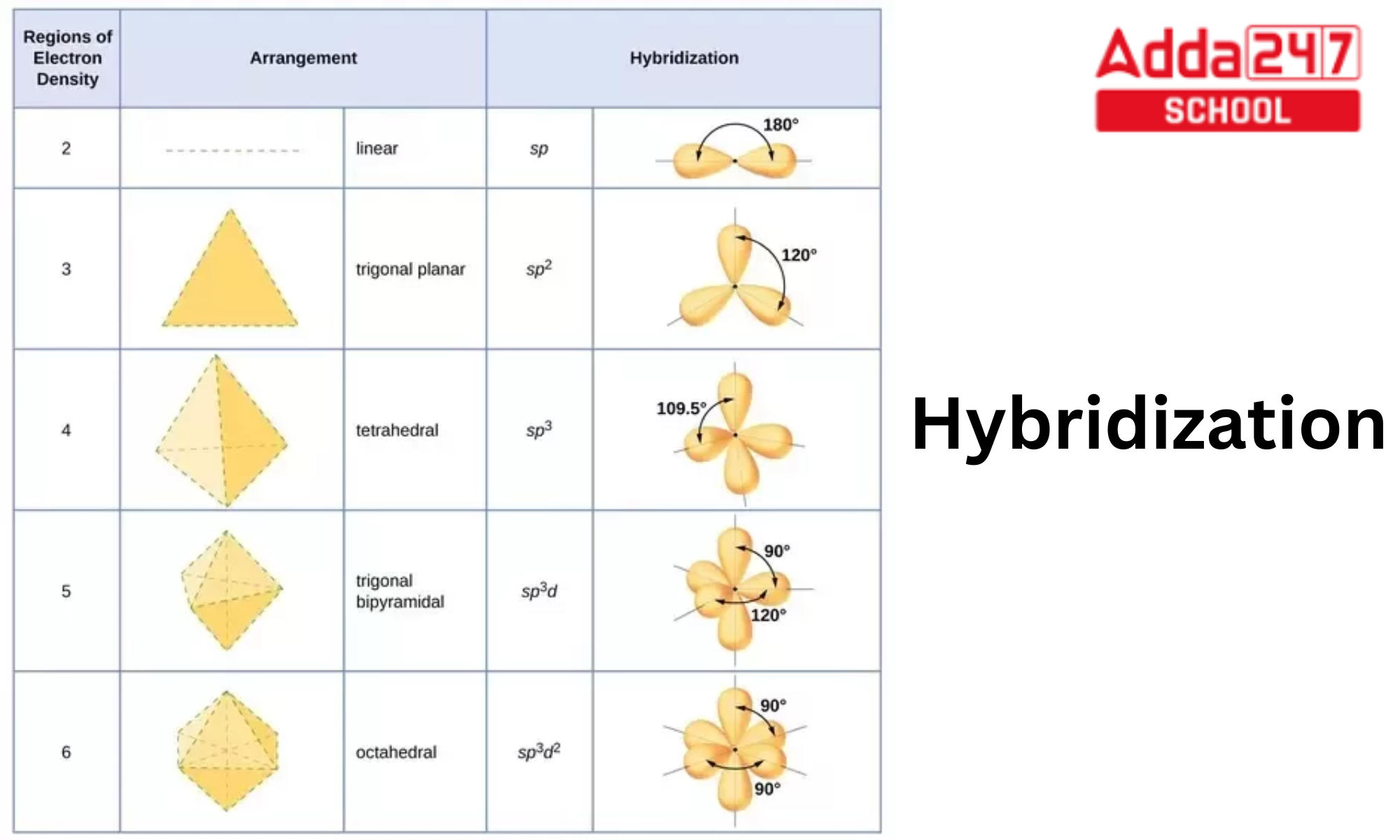
/chapter2/pages1and2/page1and2_files/sp2hyb.png)